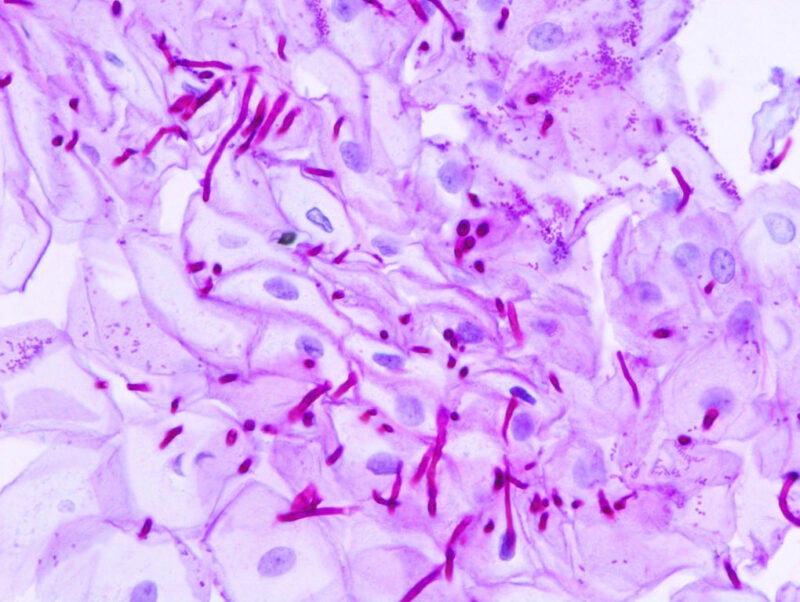
The National Institutes of Health (NIH) has awarded funding worth $9.7m to researchers at Arkansas Children’s Research Institute and Children’s Hospital of Philadelphia in the US for a clinical trial of antifungal therapy in children with uncomplicated candidemia, an invasive fungal disease.
The first-of-its-kind, randomised, controlled trial will analyse whether a briefer course of therapy can treat uncomplicated candidemia in these people.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Under the seven-year project, the scientists will assess if children with this disease show improvement following antifungal therapy for seven days.
This will aid in evaluating if the treatment can be halted at that time or if it needs to be continued for another seven days, which is the existing standard of care.
The research will also help in defining the optimal time of treatment for any invasive fungal diseases in people of any age group.
In the trial, the advantages and consequences of treating uncomplicated candidemia over a shorter period will be analysed.
It will also evaluate if a new molecular biomarker, which is already used to rapidly diagnose invasive candidiasis in children, can show if a briefer course of treatment could be a better option for patients.
The International Pediatric Fungal Network (PFN), a 55-site global research consortium, will be used for the trial.
Arkansas Children’s paediatrician in chief William Steinbach said: “This study will build a foundation for improved guidelines that allow us to treat children more rapidly, reduce the medicine they need, and get them back to thriving quickly.
“The results also will inform future trials in adults and allow us to change guidelines for care of all patients with these diseases.”





