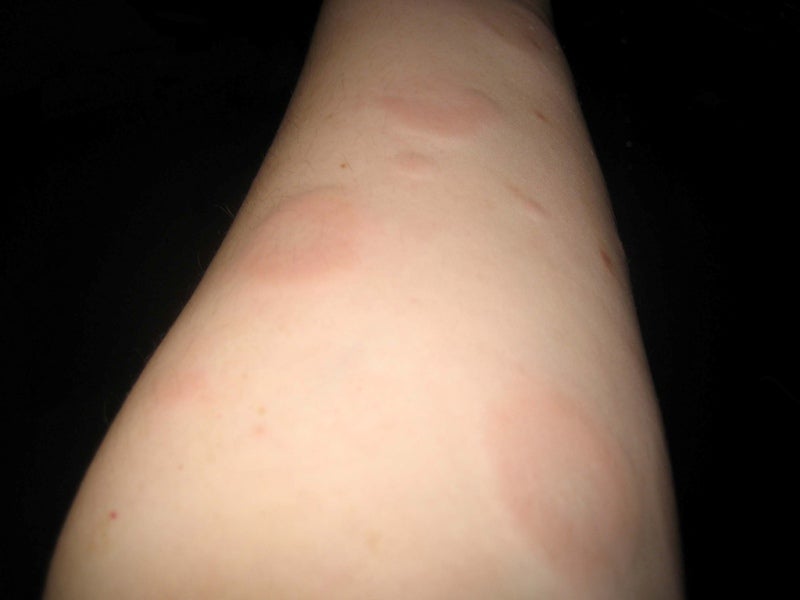
Novartis has commenced a pair of Phase III trials to evaluate the efficacy and safety of ligelizumab (QGE031) for the treatment of chronic spontaneous urticaria (CSU).
CSU is a severe and unpredictable skin condition that exhibits symptoms including spontaneous swelling.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The PEARL 1 and PEARL 2 trials are designed to determine the efficacy and safety of ligelizumab in adolescent and adult CSU patients who remain symptomatic despite H1-antihistamine treatment.
They also aim to demonstrate the better efficacy of ligelizumab over add-on CSU treatment therapy xolair (omalizumab).
The trials will follow a double-blind, placebo-controlled, parallel-group study design are expected to enrol more than 2,000 CSU patients across 48 countries including the US, Germany and Japan.
Patients will initially be randomised to receive ligelizumab dose A, ligelizumab dose B, or omalizumab 300mg every four weeks for one year.
Those who were initially randomised to placebo will be switched to receive ligelizumab dose B starting from week 24 until week 52.
Primary objective of the trial is to measure absolute change from baseline in urticaria activity score over seven days (UAS7) at week 12.
Germany’s Charité-Universitätsmedizin Allergie-Centrum-Charité Department of Dermatology and Allergy Research director and Dermatology and Allergy professor Marcus Maurer said: “CSU has a big impact on patients’ lives.
“Despite existing treatment options, too many people continue to struggle with the debilitating and potentially painful symptoms of CSU.”
The PEARL 1 and PEARL 2 trials are based on results from a Phase IIb trial of ligelizumab in comparison with xolair.
Results from the placebo-controlled trials demonstrated improvements over xolair in patients and showed a clear dose-response relationship.
Ligelizumab has also reported to have achieved rapid onset of action as well as enhanced and sustained efficacy in the patients, whose symptoms are not adequately controlled by H1-antihistamines.





