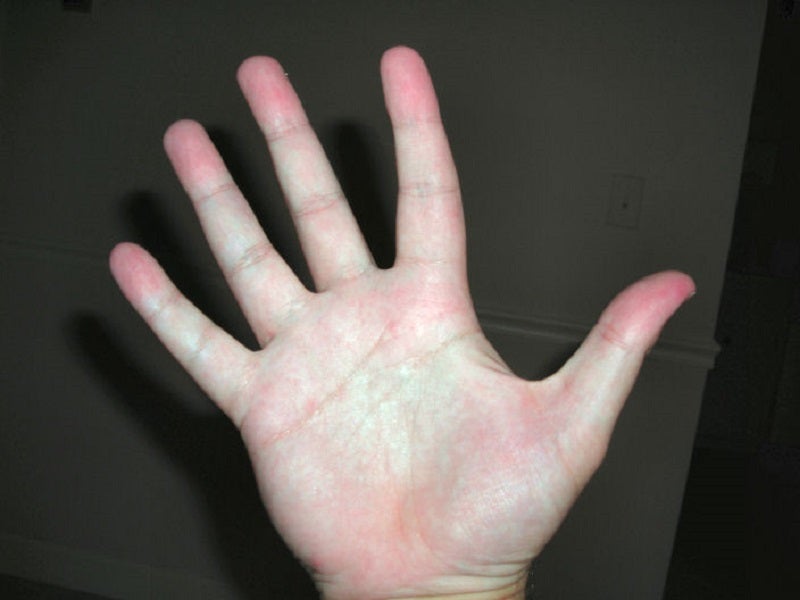
Targeted oncology supportive therapy company OnQuality Pharmaceuticals has enrolled the first participant in Part 2 of the NOVA-II Phase II clinical trial of OQL011 to manage Hand-Foot Skin Reaction (HFSR) in cancer patients.
The topical ointment will be evaluated in the patients who are receiving vascular endothelial growth factor receptor inhibitor(s) (VEGFRi) as part of their treatment.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The international multicentre, double-blinded, randomised, vehicle-controlled, dose-ranging trial will evaluate OQL011’s safety and efficacy as a topical ointment to treat VEGFR inhibitor-associated HFSR.
Those eligible for the trial must receive VEGFR inhibitor-based anticancer therapy and have HFSR severity of grade 3 or higher.
Preliminary data from the trial’s first part showed OQL011 to be safe and well tolerated without any significant adverse events.
OnQuality Pharmaceuticals chief medical officer and co-founder Hong Tang said: “Our team is delighted to start Part 2 of the NOVA-II clinical trial to address the unmet need for cancer-therapy-induced skin toxicities.
“OQL011 generated positive initial findings in Part 1 of the Phase II study, and we look forward to advancing the Part 2 study to determine the optimal dosage of OQL011 for Phase III.”
The trial’s key objectives include the assessment of the efficacy and safety of OQL011 and the identification of an optimal dosage for Phase III.
Exploring the pharmacokinetics profile of OQL011 in HSFR patients is also one of the objectives.
OQL011 acts on the key pathway of VEGFR inhibitor-associated HFSR.
HFSR is a common side effect of tyrosine kinase inhibitors, including VEGFR inhibitors that are used for the treatment of cancer.





