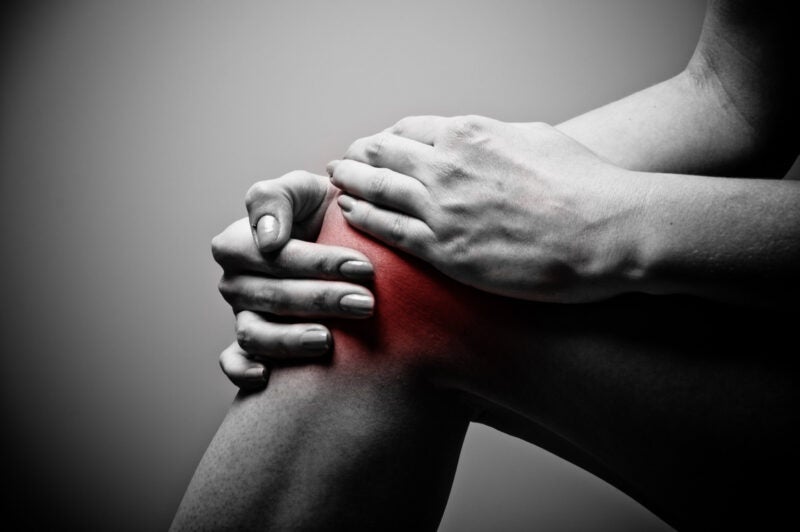
Peptilogics has dosed the first subject in the Phase Ib LOGIC-1 clinical trial of investigational product PLG0206 to treat periprosthetic joint infections (PJI) occurring following a total knee arthroplasty (TKA).
The open-label, dose-escalating trial will enrol subjects who are undergoing a debridement, antibiotics, and implant retention (DAIR) surgical procedure to treat PJI following TKA.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It will analyse the safety and effects of PLG0206 for treating PJI.
The trial subjects will be followed up for nearly one year following treatment.
A non-traditional, broad-spectrum antibiotic peptide therapy, PLG0206 can act on bacteria and persistent pathogens that form a biofilm and evade traditional antibiotics.
PLG0206 showed robust activity against Gram-positive and Gram-negative antibiotic-resistant bacteria with a reduced resistance development risk to the therapeutic in in vitro and non-clinical studies.
The therapy has received Orphan Drug and Fast Track designation from the Food and Drug Administration to treat PJI.
Peptilogics founder and CEO Jonathan Steckbeck said: “As we continue to reimagine the future of peptide therapeutic discovery, it is incredibly promising to have reached this milestone with our first programme.
“PLG0206 has demonstrated potential best-in-class, rapidly bactericidal, broad-spectrum activity against a variety of biofilm pathogens in both in vitro and non-clinical studies, and safety in a previously completed Phase I study in healthy volunteers.”
PJI is the most prevalent complication of joint replacement surgery, with patients needing an average of three surgeries for treatment, if diagnosed with PJI.
With a recurrence rate of up to 60% and a five-year death rate of 25%, the infection currently has no effective treatment.





