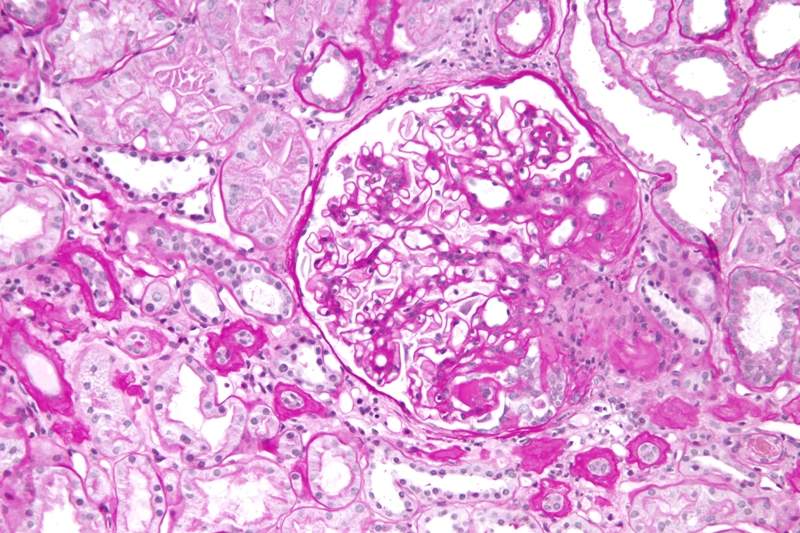
Retrophin has enrolled the first patient in the DUPLEX Study, a Phase III clinical trial examining the long-term nephroprotective potential of sparsentan to treat focal segmental glomerulosclerosis (FSGS).
The trial will evaluate the safety and efficacy of sparsentan in FSGS, a rare kidney disorder. Around 300 patients aged 8 to 75 years will be enrolled.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
In the trial, participants will be randomised to receive either sparsentan, (including an initial dose of 400mg daily for two weeks, titrating up to a target dose of 800mg daily), or an active control dose of irbesartan.
In line with the US Food and Drug Administration (FDA) feedback, the DUPLEX Study protocol allows an unblinded analysis of at least 190 patients to be performed after 36 weeks of treatment. The interim efficacy endpoint of patients achieving a modified partial remission of proteinuria at week 36 will be examined.
Successful achievement of this endpoint is expected to serve as the basis for Subpart H accelerated approval of sparsentan in the US and Conditional Marketing Authorisation (CMA) consideration in Europe.
The primary endpoint of the DUPLEX Study is the change in slope of estimated glomerular filtration rate (eGFR) after 108 weeks of treatment.
Its secondary endpoints are the percent change in eGFR from week six to week 108, as well as the percent change from baseline in Up/C at week 36 evaluated at the final analysis.
Retrophinsenior vice-president and research and development head Bill Rote said: “Patients with FSGS currently face a progressive decline that often results in end-stage renal disease and our hope is that the DUPLEX Study will enable us to deliver a first-in-class treatment to these patients in need.
“Developed in alignment with the FDA, the design of our pivotal DUPLEX Study builds upon the promising results from the Phase II DUET Study of sparsentan and positions us to pursue Subpart H accelerated approval with the inclusion of our interim endpoint assessing modified partial remission of proteinuria.”
Retrophin expects to receive top-line data from the 36-week interim efficacy endpoint analysis from the DUPLEX Studyin the second half of 2020.





