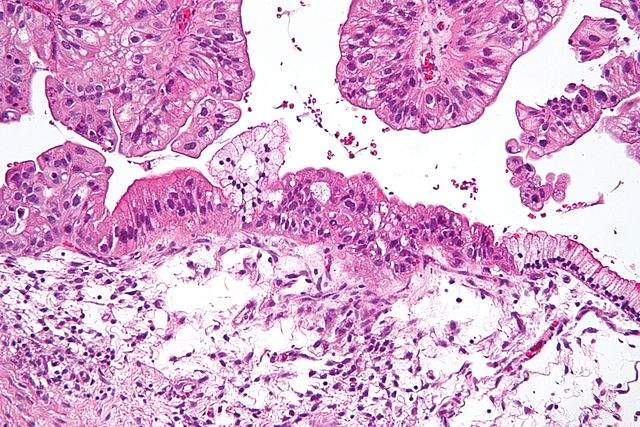
Researchers at the Moffitt Cancer Centre in Florida have used single-cell imaging of cancer cells in mice alongside mathematical models to determine the drug properties needed for efficient drug uptake.
Their findings were published in Scientific Reports and could potentially lead to the development of personalised tumour treatments.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
While existing methods for examining drug uptake are based on the idea that a tumour and its surrounding environment are homogenous, the Moffitt team is looking to update this approach to incorporate the reality of tumour cells’ differing properties. Using mathematical modelling and imaging techniques, a single cell’s uptake of a treatment was monitored and predicted. Drug characteristics and tumour properties were compared to determine the optimum conditions for effective cellular drug uptake.
The study demonstrated that the amount of a drug that binds to a cell depends on the speed at which it is diffused through a tissue, rather than its concentration. Treatments with a faster rate of diffusion were shown to bind more effectively to cells that were further from blood vessels. In comparison, drugs with a slow diffusion rate bound to cells that were closer to blood vessels and were more tightly packed. Drugs that were released more quickly were also shown to bind more effectively to cells with differing levels of drug receptors.
These findings imply that changing the properties of a drug or the way in which it is administered could improve its efficacy in targeting tumour cells.
“To treat the fast-growing cells located near the vasculature, slowly diffusing agents may be beneficial,” Moffitt Cancer Center researcher Kasia A. Rejniak said. “In contrast, for the dormant cells in poorly vascularised regions, the highly mobile agents may be preferential, or in some cancers, local injection directly to the tumour site may be beneficial.”

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataThe research attempts to tackle a problem inherent to targeted therapies. Though treatments that target specific molecules have greatly improved overall survival rates, some patients don’t respond to therapies due to the drugs’ failure to effectively reach tumour cells.
One of the main causes of a drug’s impaired efficacy is the complexity of tumours and their surrounding environments. Each tumour consists of varying cells and targeted membrane receptors, while their environment is composed of cell types with properties and densities that differ in each case. As such, therapies often cannot effectively target all of the cells in a tumour, and sometimes a cell’s imperfect exposure to the drug allows resistance to build up.
“Clinical success or failure of targeted therapy depends heavily on whether the drug molecules are able to reach all tumour cells and engage with their molecular targets to invoke the desired therapeutic effect,” Rejniak said.
Personalised medicine (or precision medicine) has been an aim of drug developers and researchers for many years, though it is only recently gaining momentum due to advancements in medical technology. It is of particular interest in the development of cancer treatments, as while tumours are generally classed in relation to the area of the body they are found in, precision medicine specifies tumours according to their genetic make-up. This offers the possibility of more targeted and cost-effective treatments that can respond to the genetic changes specific to each patient’s cancer.
There is the hope that in future, genetic tests will offer insight into which treatment a patient’s tumour will respond to, saving time and money, and avoiding the physical and emotional burden of unnecessary surgeries on patients.





