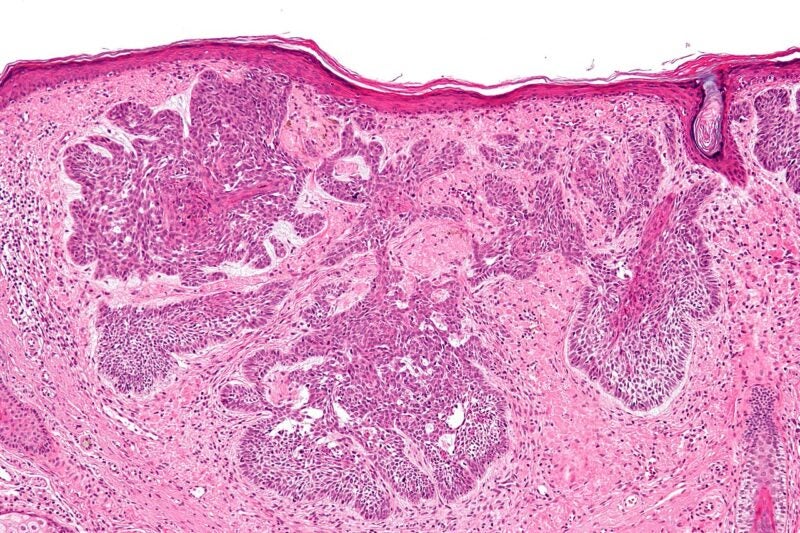

Sirnaomics has reported that a cohort, which received a 180μg dose level of STP705 in the Phase II clinical trial, attained a 100% complete response (CR) to treat cutaneous basal cell carcinoma (BCC), a kind of nonmelanoma skin cancer (NMSC).
The open-label, dose-escalation trial is intended to assess the safety, tolerability and efficacy of varying dose levels of STP705 given as a localised injection in BCC patients.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
According to the trial findings from five subjects who received the treatment in the 180μg dose cohort, enhanced or steady cosmetic results were reported.
In addition, the therapy had an exceptional safety profile without any adverse events as well as no substantial cutaneous skin reactions.
The trial will extend the number of participating cohorts to assess various doses of STP705.
The company plans to report the final data from the trial in the first quarter of next year.
A small interfering RNA (siRNA) therapy, STP705 comprises two siRNA oligonucleotides that act on the TGF-β1 and COX-2 messenger ribonucleic acid (mRNA) expression, respectively.
It has obtained investigational new drug application approvals from the US Food and Drug Administration and the Chinese National Medical Products Administration for treating NMSC, cholangiocarcinoma and hypertrophic scar.
At present, the product candidate is being analysed in seven trials for various indications such as squamous cell carcinoma in situ (isSCC), facial isSCC, BCC, keloid scarring, liver cancer (basket), hypertrophic scar, and for medical cosmetology therapy.
Sirnaomics founder, board chairman, executive director, president and CEO Dr Patrick Lu said: “The latest results from the Phase II clinical study of STP705 for BCC treatment, showing an incredible efficacy without any drug-related AEs and SAEs, further validated the broad potential of this drug candidate for treatment of non-melanoma skin cancers and beyond.
“Based on the successes of both BCC and isSCC clinical studies, Sirnaomics is spearheading the development of the novel polypeptide-based siRNA therapeutics for various types of cancers.”
Earlier, the company dosed the first subject in the Phase I/II trial of STP705 for treating facial isSCC.
Cell & Gene Therapy coverage on Clinical Trials Arena is supported by Cytiva.
Editorial content is independently produced and follows the highest standards of journalistic integrity. Topic sponsors are not involved in the creation of editorial content.


