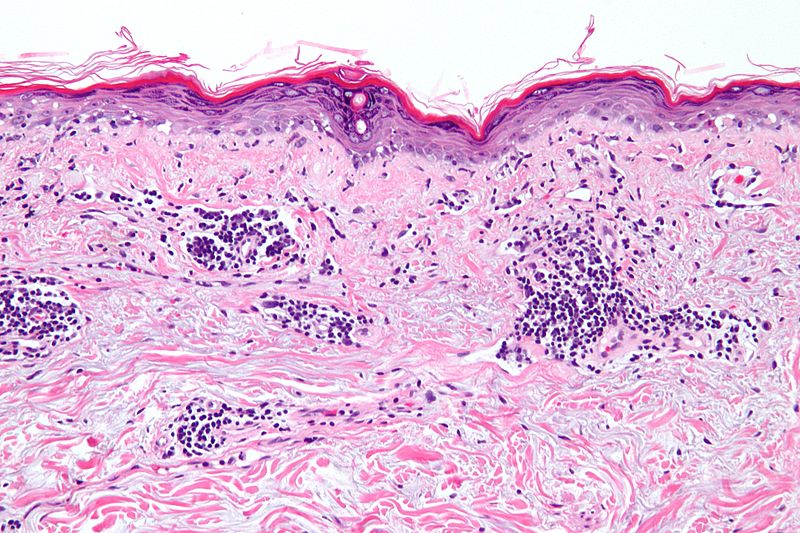
AstraZeneca has reported positive results from the Phase III Treatment of Uncontrolled Lupus via the Interferon Pathway (TULIP) 2 trial for anifrolumab to treat systemic lupus erythematosus (SLE).
The drug has achieved a significant reduction in SLE disease activity versus placebo, with both arms receiving standard of care.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
AstraZeneca said that the disease reduction was measured using the British Isles Lupus Assessment Group based Composite Lupus Assessment (BICLA) at week 52.
The randomised trial, which enrolled a total of 373 eligible patients, has been designed to measure the safety and efficacy of anifrolumab as a treatment for adults identified with moderate-to-severe SLE.
The results demonstrated that the safety and efficacy profile of anifrolumab was consistent with previous trials.
According to the company, the positive BICLA response in TULIP 2 was consistent with a pre-specified analysis of the previous Phase III TULIP 1 trial, which did not achieve positive results of SLE Responder Index 4 (SRI4).
BioPharmaceuticals R&D executive vice-president Mene Pangalos said: “Systemic lupus erythematosus is a debilitating autoimmune disease but only one new treatment has been approved in the last 60 years.”
SLE is an autoimmune disease where the immune system attacks healthy tissue in the body rather than primarily targeting viruses or other foreign invaders.
TULIP 2 trial principal investigator Professor Eric Morand said: “These exciting results from the TULIP 2 trial demonstrate that by targeting the type I interferon receptor, anifrolumab reduced disease activity in patients with systemic lupus erythematosus.”
Last September, AstraZeneca reported negative top-line results from the TULIP I trial after the study failed to meet its primary endpoint.





