French biotech Valneva has announced positive results from its Phase I clinical trial of vaccine candidate VLA15 for Lyme disease.
The drug candidate is a multivalent protein subunit vaccine that works through targeting the outer surface protein A (OspA) of Borrelia burgdorferi, the bacteria that cause Lyme disease. Researchers hope it will provide active, prophylactic immunisation against Lyme disease in patients over the age of two.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
VLA15 is designed to increase the production of antibodies that target Borrelia, preventing its spread from infected Ixodes ticks to humans after a bite.
The safety and tolerability of VLA15 at various dose levels and formulations was investigated in the trial. The study enrolled 179 subjects under the age of 40, none of whom had been previously infected with Borrelia burgdorferi. Patients were randomised into six treatment groups and received one of three dose levels either in an alum adjuvanted formulation or without adjuvant.
Immunogenicity was also monitored for the different dose groups, measured through identifying IgG antibodies against the six most prevalent serotypes of Lyme borreliosis in the US (ST1) and Europe (ST1 to ST6) present in the vaccine. This interim analysis for the primary and secondary endpoints included safety and immunogenicity data up to Day 84.
The trial’s primary objective of ensuring the drug’s positive safety profile was reached. The treatment had no associated safety concerns and there were very few severe adverse effects seen in all treatment groups.
The safety profile of the doses and formulations tested is compared to other licensed lipidated recombinant vaccines or lipid-containing vaccine formulations, and supports further clinical development for all doses and formulations.
VLA15 was shown to be immunogenic in all its doses and formulations, though it was found to be most immunogenic in adjuvanted treatment groups as compared to non-adjuvanted treatment mMthe. OspA-specific IgG antibody responses were induced in all treatment arms, tested against OspA serotypes. Seroconversion rates for the highest, adjuvanted dose group ranged from 71.4% to 96.4% for the different OspA serotypes. This is the group considered preferable for further development, with a Phase II trial of the treatment anticipated to start in the latter half of 2018.
This next phase will be conducted in Lyme-endemic regions and is to include subjects who have been previously infected with Borrelia burgdorferi.
VLA15 is the only vaccination programme against Lyme disease currently under clinical development. It was granted fast-track approval by the US Food and Drug Administration (FDA) in July 2017.
“We are extremely pleased to report a successful first human trial for our vaccine candidate against Lyme disease, a severe infection which affects an increasing number of people each year,” Valneva CEO Wolfgang Bender said. “We look forward to providing access to an effective prevention against a disease that is too often underdiagnosed, leaving many infected people with no or inadequate treatment and resulting in a heavy public health and economic burden.”
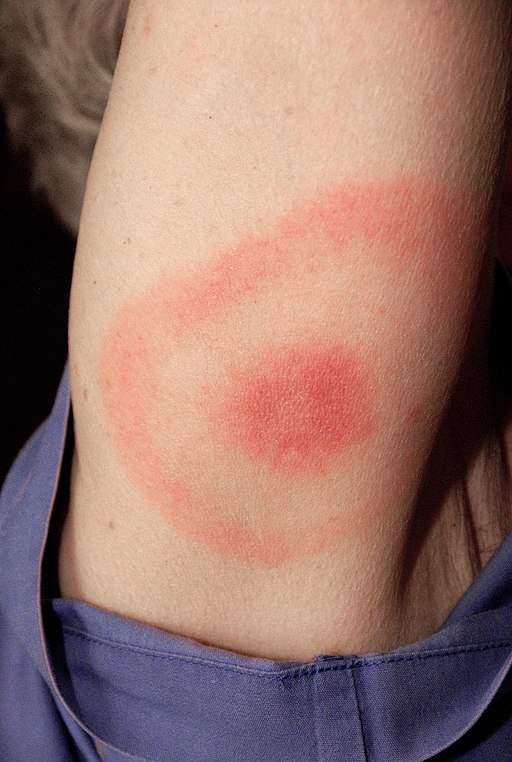
Lyme disease is a systemic infection considered to be the most common vector-borne illness in the northern hemisphere, and is the most common infection in the US following a pest bite. According to the Centers for Disease Control and Prevention (CDC), around 400,000 people in the US are diagnosed with Lyme disease per year, with a further 200,000 cases in Europe. Reported cases of the disease in the UK have been rising for the past 30 years, and in 2017 the National Health Service (NHS) estimated that 2,000 to 3,000 cases of the disease are confirmed each year, ten times more than in 2001.
The global market for a vaccine against Lyme disease is currently estimated at around €700m to €800m annually. The need for a vaccine has been increasing in recent years, with researchers warning of an imminent outbreak of the virus. Due to the world’s changing climate, Ixodes ticks have been spreading to places with no prior experience or knowledge of the disease, extending across the US, throughout Europe and forested parts of Asia.
As many countries are unaware of or unfamiliar with the condition, early symptoms are often misdiagnosed or overlooked. People often experience flu-like symptoms such as fatigue, headaches and fever, though some also report an erythematous rash and psychological effects such as depression.
If left untreated, the illness can start to affect the patient’s joints, heart and nervous system. In extreme cases, patients can experience disabling symptoms such as permanent impairment of motor or sensory function in the lower extremities.





