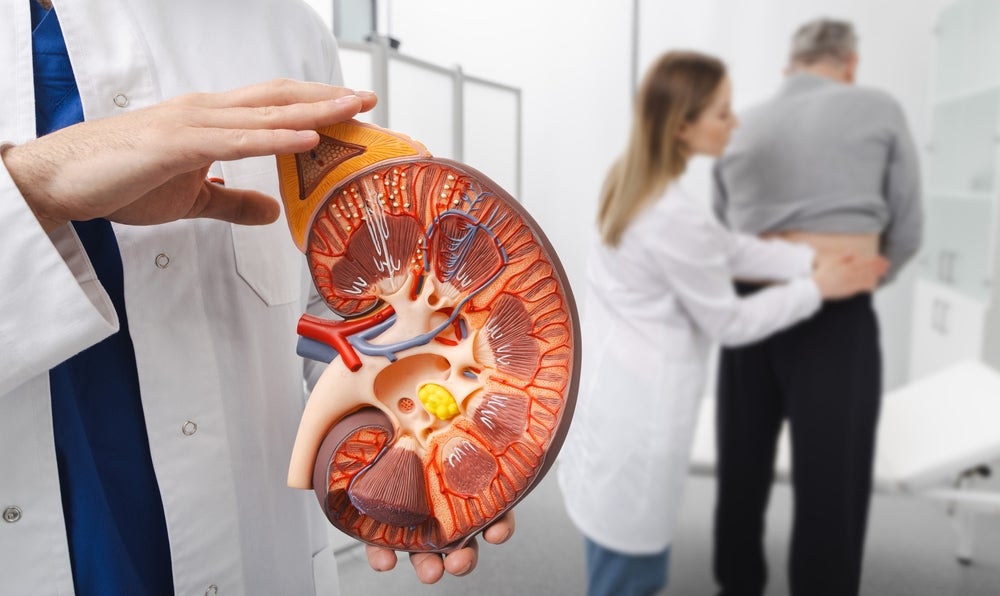
Roche is heading to regulators after its blockbuster kidney disease candidate, Gazyva (obinutuzumab), met its primary endpoint during a Phase III trial in membranous nephropathy.
In the 104-week, head-to-head MAJESTY study (NCT04629248), Gazyva bested Astellas Pharma’s commonly prescribed off-label immunosuppressant, Prograf (tacrolimus), in membranous nephropathy. The CD20-targeting monoclonal antibody (mAb) triggered complete remission (CR) in a significantly higher proportion of patients over Prograf at the two-year mark – meeting its primary endpoint.
Meanwhile, analysis of key secondary endpoints revealed that patients treated with Gazyva experienced a statistically significant increase in overall remission classed as either complete or partial at week 104. This same effect was observed on CR at week 76.
Gazyva’s safety and tolerability profile in the study also fell in line with the drug’s activity in previous trials, with no new safety signals identified.
While Roche did not share any further data from the MAJESTY study, the Swiss pharma company plans to present data from the trial at an upcoming medical meeting. Roche will also share these data with US and EU regulators, which could see Gazyva – known as Gazyvaro in the EU – become the first drug to gain approval in membranous nephropathy.
This will be a positive step forward for Roche, which is looking to expand Gazyva’s legacy outside of the cancer and lupus nephritis (LN) markets amid the looming expiry of its key patents, which begin in early 2030. To achieve this, the company is evaluating Gazyva in both paediatric nephrotic syndrome (PNS) and systemic lupus erythematosus (SLE) through the Phase III INShore and ALLEGORY (NCT05627557; NCT04963296) studies, respectively.
The company’s efforts appear to be paying off, as Gazyva has already met its primary trial endpoints in both studies.
Pioneering the membranous nephropathy market
Membranous nephropathy is a chronic autoimmune disease characterised by progressive damage to the kidney’s glomeruli, which can result in reduced kidney function, high blood pressure and proteinuria. Researchers estimate that the disease impacts eight to 10 people per one million globally.
No regulatory body has officially granted approval to any drug in membranous nephropathy, though doctors use several treatments – including Prograf and anti-CD20 antibody rituximab – off-label.
Other companies are looking to capture some of this emerging market, with several drugs progressing to late stages of development. This includes BeOne Medicines, which is evaluating its oral Bruton’s tyrosine kinase (BTK) inhibitor, Brukinsa (zanubrutinib), in the Phase II/III ALMOND (NCT05707377) trial. The ALMOND study will reach primary completion in October 2027, as per ClinicalTrials.gov.
Meanwhile, Biogen is evaluating intravenous medication, felzartamab, in the Phase III PROMINENT (NCT06962800) trial, with a readout anticipated in 2029.
GlobalData, parent company of Clinical Trials Arena, forecasts that Gazyva’s global sales will peak at $1.8bn in 2030.