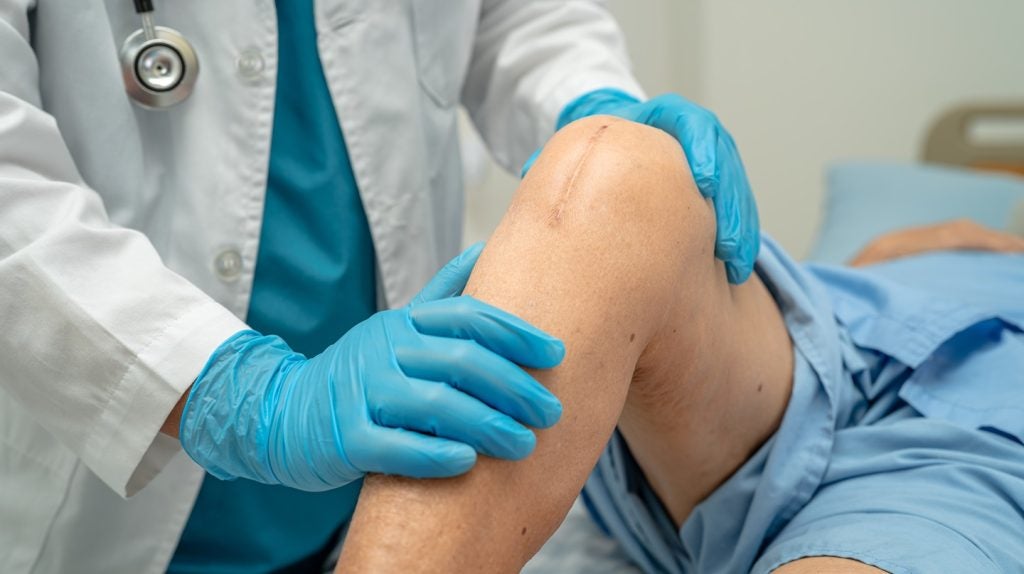
Sirius Therapeutics and CRISPR Therapeutics have dosed the first patient in a Phase II trial of SRSD107 for thromboembolic disorders in Europe.
This trial aims to investigate SRSD107 in preventing venous thromboembolism (VTE) in individuals who are undergoing total knee arthroplasty (TKA).
The strategic collaboration between the companies focuses on developing new treatments for clotting-related and cardiovascular diseases.
SRSD107, a long-acting Factor XI (FXI) small interfering RNA (siRNA), is a key outcome of this partnership.
The multi-centre, global study, which is underway, is randomised and will assess the efficacy and safety of the therapy.
The trial's results will be pivotal in determining the therapy’s pharmacological profile and anticoagulant effects, which could lead to a new approach for minimising thrombotic risk in subjects.
CRISPR Therapeutics chief medical officer Naimish Patel said: “Until now, existing anticoagulant options have been limited by bleeding risk, frequent dosing, and complex management challenges for patients with high thrombotic risk.
“SRSD107 offers the potential to reduce pathological thrombosis while minimising bleeding risk, with sustained but reversible pharmacodynamic effects and the possibility of infrequent dosing.
“We look forward to exploring how this differentiated approach could meaningfully improve outcomes for patients in need.”
SRSD107's mechanism targets FXI, a significant contributor to pathological thrombosis, while aiming to reduce the impact on normal haemostasis.
Previous Phase I studies in China and Australia showed that SRSD107 was tolerated well, with single doses leading to sustained reductions in FXI levels and a rise in activated partial thromboplastin time.
Thrombosis, the formation of blood clots, is a major cause of ischemic stroke, myocardial infarction and venous thromboembolism.
In February 2024, Sirius Therapeutics dosed the first participant in a Phase I study of SRSD107 to prevent and treat thromboembolic disorders.