Annotated CRFs
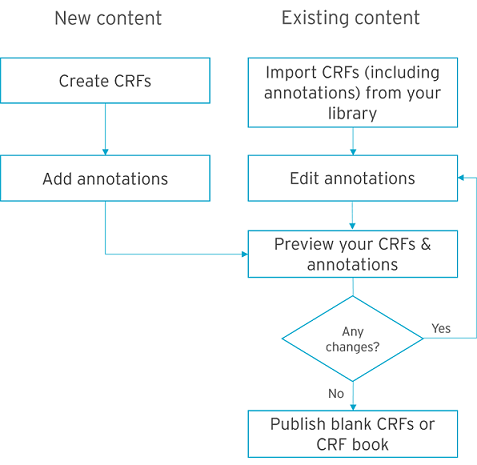

Annotating CRFs (Case Report Forms) is an important but almighty task. You need to manually create annotations for every single form, then convert your forms into PDF, which is a lot of time and resources.
However, you do not need to do it the hard way. You can see how it is done in Formedix, in the diagram above.
Once your CRF designs are created in Formedix, you can easily annotate them. We simply use your metadata to make the annotations for you. Then, just press a button to see what your CRF design looks like in PDF format. Easily make any changes. And instantly preview again until you’re done. Imagine how much time you could save, and how much more productive you could be.
Why use Formedix for SDTM annotations?
Formedix makes it much easier to create annotated CRFs with minimal effort. Gone are the days of manually creating annotated CRFs, then having to verify that it matches the collected data. Once the data acquisition forms for your study have been defined upfront, you can add annotations in your SDTM.
Then, it is one push of a button to generate a regulatory compliant SDTM annotated CRF, based on your original study design, so you know it is right. That means no more rushing to manually create your annotated CRFs when pulling together your submission.
When you have end-to-end standards, your data acquisition forms are aligned with your downstream datasets. When creating a study from your standards, everything comes along – your forms, terminologies, edit checks, datasets, and the annotations that drive the annotated CRF.
How Formedix makes annotated CRFs less of a chore
One of the main benefits of using automated annotated CRFs in Formedix is that you have detailed control over your form layouts. You decide on the font, display format, placement, colour, and type of annotations.
Types of annotation?
You can use annotations for more than mapping between fields on a form to variables in a dataset. You can create annotation notes next to fields for SAS Programmers, Data Management, and reviewers.
You can even provide other useful information, such as indicating which fields are required or hidden. Tailor annotations to fit how you want to work.
Previewing annotations
Why painstakingly create manual annotations on PDF’s, one form at a time? Let Formedix do it for you and save time and resources. Just switch on the plugin of your choice. Then, set your annotation type, display format, and font. Next, set your SDTM annotations on each field. Now you are good to go. You can easily preview what your annotations will look like, so you know they are correct.
Reusing annotations
You can reuse your standards and studies, including annotations, so you don’t have to do them again. That gives you consistency across studies, standards, and in your SDTM. It also reduces the margin for error.
