Gazyva (obinutuzumab / GA101) is a monoclonal antibody indicated for the treatment of chronic lymphocytic leukaemia (CLL).
Discovered and manufactured by Roche, the drug obtained US Food and Drug Administration (FDA) approval for the treatment of CLL patients in November 2013.
FDA approval was also secured for the treatment of follicular lymphoma and advanced follicular lymphoma in February 2016 and November 2017, respectively.
When combined with chlorambucil chemotherapy, Gazyva was approved by the European Commission (EC) for the treatment of CLL patients in July 2014. It is marketed as Gazyvaro in EU and Switzerland.
Gazyvaro also received approval from the EC for the treatment of previously untreated advanced follicular lymphoma in September 2017.
Chronic lymphocytic leukaemia (CLL)
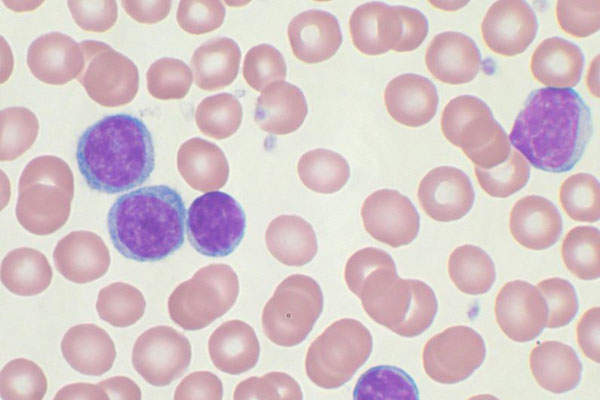
Chronic lymphocytic leukaemia (CLL) is a white blood cell cancer that originates in the bone marrow and spreads to other parts of the body.
It is a slow-growing leukaemia and is the second most common type in adults, usually affecting patients during or after middle age.
An estimated 16,000 new cases of CLL are registered every year and it was said to cause approximately 5,000 deaths in the US in 2013. The disease is responsible for approximately 10,000 deaths a year in Europe.
Gazyva’s mechanism of action against CLL
Gazyva contains a humanised monoclonal antibody, which works with the body’s immune system to target the CD20 molecule found on B-cells, which are responsible for causing blood cancer.
The drug is available as an injection for intravenous administration.
Clinical trials on Gazyva
The FDA and EU approvals for Gazyva were based on results obtained from Phase III CLL11 clinical trial, which was conducted in association with the German CLL Study Group (GCLLSG).
The open-label, multi-centre, randomised, three-arm study investigated the efficacy and safety of Gazyva plus chlorambucil or MabThera/Rituxan plus chlorambucil compared with chlorambucil alone.
The study enrolled 781 patients with CLL and co-existing medical conditions. The primary endpoint of the study was progression-free survival (PFS), while the secondary endpoints included overall response rate (ORR), overall survival (OS), disease-free survival (DFS), minimal residual disease (MRD) and safety profile.
The results demonstrated that patients administered with Gazyva in combination with chlorambucil chemotherapy showed an 84% reduction in the risk of disease worsening or death in comparison with those who received chlorambucil alone.
The median PFS in Gazyva in combination with chlorambucil chemotherapy administered patients was 23 months in comparison with 11 months in patients who received chlorambucil alone.
Patients administered with Gazyva plus chlorambucil exhibited a 75.9% overall response rate compared to 32.1% with chlorambucil alone. The study also found that approximately 27.8% of people who received Gazyva in combination with chlorambucil achieved a complete response compared to 0.9% with chlorambucil alone.
Common adverse events (AE) encountered by patients administered with Gazyva in combination with chlorambucil chemotherapy during the clinical trial included infusion-related reactions, low-platelet count and low-count of certain types of white blood cells.
FDA approval of Gazyva for previously treated follicular lymphoma is based on results obtained from open-label, multicentre, randomised, two-arm Phase III trial GADOLIN. The study evaluated 413 indolent non-Hodgkin lymphoma (iNHL) patients. Of these, 321 patients were diagnosed with follicular lymphoma.
The EU and FDA approvals for previously untreated advanced follicular lymphoma is based on results derived from the open-label, multi-centre, randomised two-arm Phase III GALLIUM study evaluating 1401 patients with previously untreated iNHL, including 1202 follicular lymphoma patients.
Marketing commentary
Gazyva is the first breakthrough therapy designated drug approved by the FDA. The drug is marketed in the US by Genentech, a member of Roche Group, in partnership with Biogen Idec.
Roche is a leading biotech company that is headquartered in Basel, Switzerland. The group focuses on healthcare research and manufactures medicines in oncology, infectious diseases, inflammation, metabolism and neuroscience. It also provides diagnostic tools and employs around 85,000 people worldwide.
Related content
Xalkori – Treatment for Non-Small Cell Lung Cancer
Xalkori (Crizotinib) is an oral drug indicated for the treatment of non-small cell lung cancer (NSCLC).
Sutent (Sunitinib Malate) – Treatment for Pancreatic Neuroendocrine Tumours, United States of America
Sutent (sunitinib malate) is an oral multikinase inhibitor which is indicated for the treatment of progressive, well-differentiated pancreatic neuroendocrine tumours (NET).