Libtayo (cemiplimab-rwlc) is an advanced fully human monoclonal antibody indicated for the treatment of both advanced cutaneous squamous cell and basal cell carcinomas and advanced non-small cell lung cancer (NSCLC).
Developed by Regeneron using its proprietary VelocImmune® technology, Libtayo is approved in the US and is being jointly marketed by Regeneron and Sanofi Genzyme as part of a collaboration agreement between the two companies.
Approvals on Libtayo
The US Food and Drug Administration (FDA) granted breakthrough therapy designation for Libtayo in September 2017. The biologics license application (BLA) for Libtayo was accepted for priority review in April 2018. The drug was approved by the FDA for the treatment metastatic or locally advanced cutaneous squamous cell carcinoma (CSCC) in September 2018.
Regeneron and Sanofi Genzyme submitted a marketing authorisation application (MAA) to the European Medical Agency (EMA) for Libtayo, which was accepted for review in April 2018. The review process was completed in the first half of 2019.
Libtayo received approval from the European Commission (EC) for use in adult patients with metastatic or locally advanced CSCC in July 2019.
In February 2021, Libtayo (cemiplimab-rwlc) received approval from the FDA as the first immunotherapy indicated for treating advanced basal cell carcinoma (BCC) who are previously treated with a hedgehog pathway inhibitor (HHI) or for whom an HHI is not acceptable.
The drug also received approval as first-line treatment for patients with advanced NSCLC with high PD-L1 expression in the same month. Libtayo (cemiplimab) received approval from the European Commission for NSCLC treatment in June 2021.
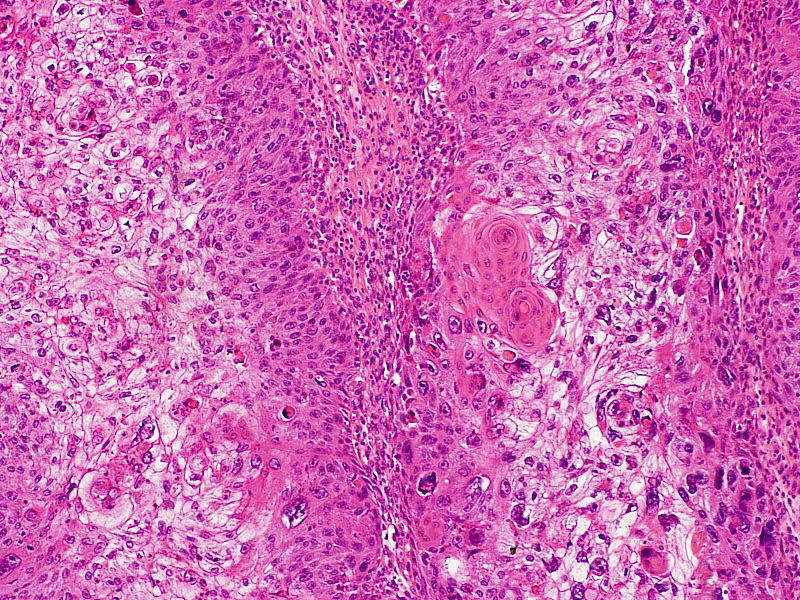
Cutaneous squamous cell carcinoma causes and symptoms
CSCC is a type of skin cancer arising from the uncontrolled growth of cells in the upper layers of the skin, often characterised by a presence of persistent scaly patches, open sores, warts and elevated growths with a central depression, which may crust and bleed.
The most common causes of CSCC are long-term exposure to ultraviolet (UV) radiation from sunlight and the use of indoor tanning. However, it can also be caused by skin injuries such as scars, long-standing sores, burns, ulcers, chronic infections, skin inflammation and X-ray exposure.
CSCC is the second most common cancer in the US, with approximately 700,000 cases reported a year. It is also responsible for causing 7,000 deaths annually in the US.
BCC is the most prevalent kind of skin cancer, in which a tiny percentage of tumours can progress and penetrate deep into surrounding tissues or spread to other regions of the body, making treatment more challenging.
NSCLC accounts for approximately 84% of all lung cancers, with 75% of these cases identified in advanced stages and an estimated 25% to 30% of cases tested positive for PD-L1 in ≥50% of tumour cells.
Libtayo’s mechanism of action
Libtayo is a recombinant human immunoglobulin G4 (IgG4) monoclonal antibody that binds to programmed death receptor-1 (PD-1), responsible for regulating T-cell response.
The drug blocks PD-1 interaction with PD-L1 and PD-L2 ligands and clears PD-1 pathway-mediated immune suppression, including the anti-tumour immune response, resulting in increased tumour surveillance and decreased tumour size.
Libtayo is available for intravenous use in the form of a sterile and opalescent solution.
Clinical trials on Libtayo
FDA approval for Libtayo for CSCC comes from data of Phase II trial, EMPOWER-CSCC-1 (Study 1540) and a Phase I trial (Study 1423). A total of 108 patients enrolled in the studies, with 82 in Study 1540 and 26 in Study 1423.
Both trials were open-label, multi-centre, non-randomised, multi-cohort studies that excluded patients with autoimmune diseases and those that underwent prior treatment with anti–PD-1 / PD-L1-blocking antibodies or other immune checkpoint inhibitor therapy.
Patients were administered with 3mg/kg of Libtayo every two weeks for up to 48 weeks in Study 1423 or up to 96 weeks in Study 1540. The primary endpoint of the study was objective response rate (ORR), or the percentage of patients that experienced either partial shrinkage or complete disappearance of tumours post-treatment.
Patients administered with Libtayo showed an ORR of 47.2% with a significant reduction in tumours. The most common adverse events reported in trials were fatigue, rash and diarrhoea.
Libtayo’s approval for BCC by the FDA was based on an open-label, multi-centre, non-randomised Phase II clinical trial conducted in 132 unresectable locally advanced BCC or metastatic BCC patients with the primary endpoint of objective response rate (ORR). The drug achieved confirmed ORR of 21% in metastatic BCC group and 29% in locally advanced BCC group.
The drug’s approval for NSCLC was supported by a Phase III clinical trial conducted in 710 patients. Libtayo reduced the risk of death in patients by 32% compared to chemotherapy.