Lipaglyn™ (saroglitazar) is a dual peroxisome proliferator-activated receptor (PPAR) agonist indicated for the treatment of hypertriglyceridaemia in Type 2 diabetes (T2D).
The drug is also approved to treat liver conditions such as non-alcoholic steatohepatitis (NASH), non-alcoholic fatty liver disease (NAFLD) and primary biliary cholangitis (PBC).
Lipaglyn was discovered and developed by Zydus Lifesciences (formerly Zydus Cadila), an India-based pharmaceutical company. It is available as tablets of 4mg for oral administration.
Zydus has signed co-marketing agreements for the drug in India with Lupin and Torrent Pharmaceuticals for the treatment of NAFLD and NASH.
Lupin markets the drug under the brand name LINVAS® and Zydus markets it under the brand name VORXAR®.
Zydus continues to market the drug under the brand names Lipaglyn and Bilypsa®.
Regulatory approvals for Lipaglyn
In 2012, Zydus submitted a new drug application for Lipaglyn’s approval to the Drug Controller General of India (DCGI). The company received marketing approval for Lipaglyn from the DCGI in June 2013 for treating diabetic dyslipidaemia in T2D patients.
The drug is also approved by the DCGI for the treatment of T2D as an add-on therapy with Metformin and for the treatment of NAFLD and NASH.
Zydus received marketing approval for Lipaglyn in Mexico in November 2017 to treat dyslipidaemia in T2D patients and hypertriglyceridemia in T2D patients whose conditions could not be controlled by statins.
In December 2020, saroglitazar Mg was given fast-track designation by the US Food and Drug Administration to treat individuals with PBC. The drug also received orphan drug designation in January 2021.
Saroglitazar Mg received orphan drug designation from the European Medicines Agency in July 2021 for the treatment of PBC.
Hypertriglyceridaemia in T2D causes and symptoms
Hypertriglyceridaemia, also known as diabetic dyslipidaemia, is a condition whereby a person with diabetes has high blood lipid levels or triglycerides.
The elevated level of triglycerides is one of the major risk factors associated with cardiovascular diseases (CVD).
India is estimated to have approximately 77 million people with diabetes, with the majority of them suffering from diabetic dyslipidaemia. An estimated 85% of people with diabetes worldwide suffer from diabetic dyslipidaemia.
Studies have shown that around 30% of deaths in the world are caused by CVD, and diabetes is one of the major causes associated with CVD.
PBC causes and symptoms
PBC is a liver disease caused by progressive destruction of the bile ducts in the liver. This results in decreased bile flow, a condition known as cholestasis.
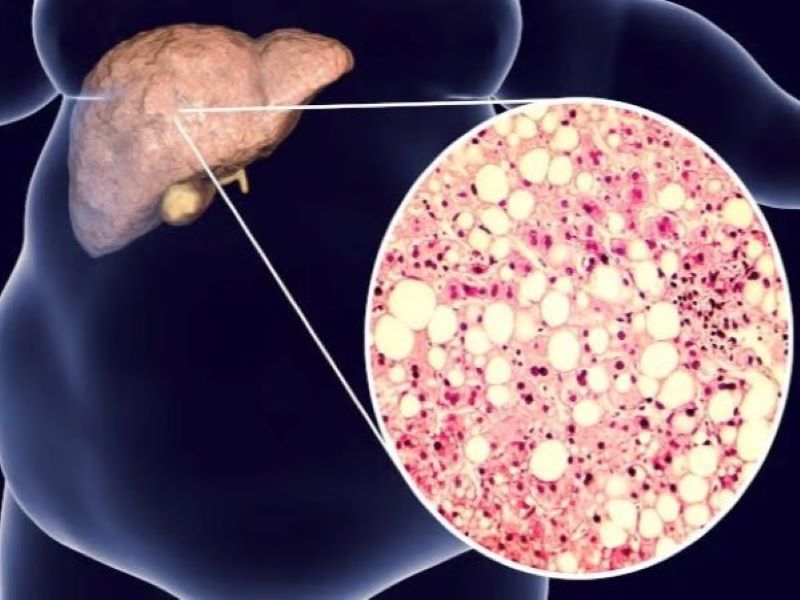
NAFLD causes and symptoms
NAFLD is a chronic, worsening liver disorder characterised by the build-up of fat in the liver in people who drink no alcohol or only negligible amounts.
It is commonly associated with metabolic risk factors including overweight/obesity, diabetes mellitus, hypertension and dyslipidaemia.
Over time, NAFLD may advance to NASH, and subsequently to cirrhosis and end-stage liver failure.
Lipaglyn (saroglitazar) mechanism of action
Lipaglyn contains two main classes of PPAR agonists: PPARα (alpha) and PPARγ (gamma). The drug includes both lipid and glucose-lowering effects in a single molecule. It lowers high blood triglycerides and blood sugar, as well as improves insulin resistance.
In NASH models, the drug was found to act on all disease components, such as steatosis, ballooning, inflammation and fibrosis.
Clinical trials on Zydus’s Lipaglyn (saroglitazar)
The approval of Lipaglyn for the diabetic dyslipidaemia indication was based on results from clinical studies, which had been conducted for more than eight years. The studies evaluated the drug’s efficacy, safety, pharmacokinetics and pharmacodynamics.
Phase I clinical trials on Lipaglyn were conducted in 2005, and Phase II studies were completed in 2006. The Phase III clinical trials were conducted between 2008 and 2011.
The first Phase III clinical trials on Lipaglyn compared a 4mg dose of the drug with pioglitazone, a similar drug developed by Takeda.
Results from the study demonstrated that patients who were administered 4mg of Lipaglyn showed a reduction in low-density lipoprotein cholesterol and triglycerides and an increase in high-density lipoprotein cholesterol. The study also showed that Lipaglyn-treated patients showed a reduction in fasting plasma glucose and glycosylated haemoglobin.
The second Phase III clinical trial on Lipaglyn was conducted to evaluate diabetic dyslipidaemia patients whose condition had been insufficiently controlled with statin therapy. Results from the study showed that patients treated with Lipaglyn demonstrated a pronounced beneficial effect on both the lipid and glycaemic parameters.
Lipaglyn was well-tolerated, and its safety profile was shown to be better than that of its comparator drugs in both trials. The drug was also found to have a non-renal route of elimination and did not show adverse events such as oedema, weight gain, myopathies, or derangement of liver or kidney functions, thus making it safe and efficacious.
The Phase III clinical trial on saroglitazar Mg for the treatment of T2D as an add-on therapy with metformin evaluated the drug’s safety and efficacy against those of pioglitazone. The trial met the primary endpoint as HbA1c levels reduced by 1.34g/decilitre (dL) with saroglitazar 2mg and 1.49g/dL with saroglitazar 4mg compared with 1.47g/dL with pioglitazone 30mg.
Additional clinical trials
Various other clinical trials are being carried out to evaluate the safety, tolerability and effectiveness of saroglitazar Mg in patients with NASH and PBC.
In Phase II trials involving NAFLD patients, saroglitazar Mg improved liver enzymes and demonstrated favourable effects on lipid and glycaemic indices.
The drug also yielded positive data in the Phase III biopsy-driven EVIDENCES II study, which compared 4mg saroglitazar with placebo in participants suffering from NASH.
In the EVIDENCES III trial, another Phase III biopsy-driven study, saroglitazar Mg 4mg demonstrated a significant reduction in liver fat, liver enzymes and overall disease activity compared to placebo in Indian patients with NASH.
Saroglitazar Mg achieved the primary endpoint in the EVIDENCES IV trial conducted in patients with NAFLD and NASH in the US. It showed a 44.39% reduction in alanine aminotransferase from baseline at 16 weeks apart from achieving reductions in liver fat content, insulin resistance and atherogenic dyslipidaemia.
The EPICS Phase II study to evaluate the safety, tolerability and efficacy of saroglitazar Mg in patients with PBC showed that the drug was well-tolerated and resulted in significant improvements in alkaline phosphotase (ALP) levels.
The EPICS-III Phase II(b)/III clinical trial was initiated in December 2021 to evaluate the safety and efficacy of saroglitazar Mg in patients with PBC. The study showed that saroglitazar 2mg and 4mg was well-tolerated and resulted in rapid and consistent ALP improvements.
The EVIDENCES VII Phase IIA trial was conducted to evaluate saroglitazar Mg’s ability to treat NAFLD in women with polycystic ovary syndrome in the US and Mexico. Saroglitazar at a daily dose of 4mg led to marked improvements in liver fat content, transaminase levels and lipid abnormalities in the trial.
In the EVIDENCES VIII Phase III clinical trial in patients with NAFLD, saroglitazar 4mg led to an improvement in the liver fat content, as measured by Magnetic Resonance Imaging-Proton Density Fat Fraction and an improvement in the liver enzymes and liver stiffness measured by Fibroscan®.
The EVIDENCES-X is a global pivotal Phase II(b) clinical trial being conducted to evaluate the safety and efficacy of saroglitazar Mg in patients with NASH. The study’s primary endpoint is the resolution of steatohepatitis with no worsening of fibrosis.