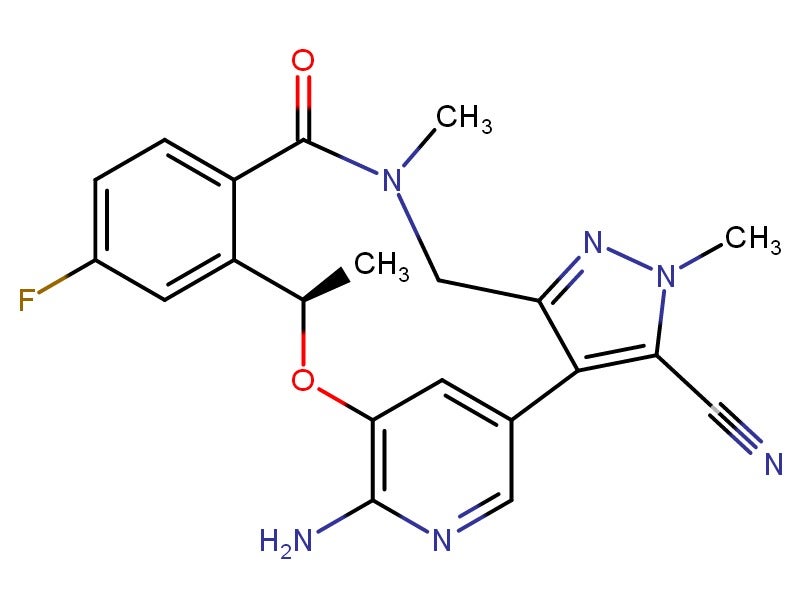
Lorbrena® (lorlatinib) is an anaplastic lymphoma kinase (ALK) inhibitor indicated as a second or third-line treatment of advanced non-small cell lung cancer (NSCLC). The drug was discovered and developed by Pfizer.
The US Food and Drug Administration (FDA) accepted the new drug application (NDA) for Lorbrena in February 2018 and granted priority review status.
Pfizer also submitted regulatory applications for the drug to the European Medicines Agency and the Japan Pharmaceutical and Medical Devices Agency, which was accepted for review during the same period.
The FDA approved the drug for treating ALK-positive metastatic NSCLC patients in November 2018. The drug is also approved in Japan to treat ALK fusion gene-positive advanced and / or recurrent NSCLC.
NSCLC causes and symptoms
Lung cancer occurs due to the uncontrolled growth and spread of the lung cells, leading to the formation of cancer tumours. There are two types of lung cancers, NSCLC and small cell lung cancer.
NSCLC is the most common type of lung cancer, accounting for approximately 80% to 85% of lung cancer cases worldwide.
Smoking tobacco is the most common cause of NSCLC development. Other factors include second-hand smoke, exposure to air pollutants such as asbestos, chemicals, and diesel exhaust.
Some of the common symptoms of the disease include long-lasting coughing that gets worse with time, abnormal change in voice, chest pain, wheezing, weight loss, and breathlessness.
Lorbrena’s mechanism of action
Tumour complexity and treatment resistance are the major causes of disease progression in patients with ALK-positive metastatic NSCLC. The brain is the most common site of tumour progression in the condition.
Lorbrena® blocks mutations of the ALK enzyme, especially those that cause treatment resistance. It is also specifically designed to penetrate the blood-brain barrier.
It is administered once a day as an oral drug available in 0.25mg and 100mg strength tablets.
Clinical trials on Lorbrena
The FDA approval of Lorbrena® was based on data from a phase I/II clinical study named B7461001. The non-randomised, dose-ranging and activity-estimating, multi-cohort, multi-centre study enrolled 295 patients with ALK-positive or ROS1-positive metastatic NSCLC.
A total of 215 patients with ALK-positive metastatic were divided across different subgroups on the basis of their prior treatment. The patients were administered with one 100mg daily dosage of Lorbrena® for a median duration of 12.5 months.
The primary endpoint of the trial was overall response rate (ORR), intracranial ORR, and duration of response. The ORR was 48%, including 44% partial response and 4% complete response. An estimated 57% of the patients treated with Lorbrena® had prior treatment with more than one ALK tyrosine kinase inhibitor.
Intracranial ORR and duration of response for CNS metastases were evaluated in a subgroup of 89 patients. The intracranial response rate in patients was 60%, while median duration of response was 19.5 months. The clinical trial included approximately 69% of patients who had a history of brain metastases.
The most common adverse events observed in the patients were oedema, peripheral neuropathy, cognitive effects, difficulty in breathing, fatigue, weight gain, joint pain, mood effects, and diarrhoea. Serious adverse reactions such as pneumonia, rise in body temperature, and respiratory failure were observed in approximately 32% of the 295 patients.