Lumakras (sotorasib) is an oral, first-in-class therapy indicated for the treatment of adult patients with KRAS G12C-mutated locally advanced or metastatic non-small cell lung cancer (NSCLC) following at least one previous systemic therapy.
Developed by US-based biopharmaceutical company Amgen, Lumakras is available as immediate-release, yellow, oblong-shaped, film-coated tablets in 120mg dosage strength.
Regulatory approvals for Lumakras
The European Commission approved Lumakras in January 2022, following the receipt of a recommendation for conditional marketing authorisation from the European Medicines Agency (EMA)’s Committee for Medicinal Products for Human Use (CHMP) in November 2021.
The Japan Ministry of Health, Labour and Welfare (MHLW) also approved the drug in January 2022.
In December 2021, Lumakras received temporary authorisation in Switzerland.
In May 2021, the therapy received accelerated approval from the US Food and Drug Administration (FDA) for the treatment of locally advanced or metastatic NSCLC with the KRAS G12C mutation. The drug was also approved for use by regulatory authorities in the UAE and Canada (as Lumakras) and the UK (as Lumykras®).
In August 2019, the FDA granted fast track designation to sotorasib for the treatment of patients with previously treated metastatic NSCLC with a KRAS G12C mutation.
The QIAGEN therascreen KRAS RGQ polymerase chain reaction (PCR) kit and the Guardant360 CDx test obtained FDA approval as a companion diagnostic for detecting NSCLC patients eligible for treatment with Lumakras.
The drug also holds breakthrough therapy designation in the US and China and has secured orphan drug designation from the FDA for KRAS G12C-positive NSCLC and colorectal cancer.
In June 2021, Amgen selected US-based oncology pharmacy company Onco360 as a speciality pharmacy provider for Lumakras.
The drug has received approval in 35 countries globally, marking a significant milestone achieved through the most advanced KRAS G12C clinical development programme.
Non-small cell lung cancer causes and symptoms
NSCLC is the most common form of lung cancer, accounting for up to 85% of all lung cancer cases.
KRAS is a member of the rat sarcoma (RAS) family and is the most frequently mutated oncogene. It is vital for the regulation of signalling pathways responsible for cell growth and division.
KRAS G12C is a single-point mutant-oncogenic form of RAS GTPase and is observed in around 13% of NSCLC patients in the US, resulting in continuous cancer cell proliferation, development and survival.
Some of the most common NSCLC symptoms include the worsening of chronic cough, cough-producing blood, wheezing, unexplained weight loss, chest pain or painful breathing, decreased appetite, and lung infections such as bronchitis and pneumonia.
Advanced-stage symptoms include bone pain, headaches or seizures due to brain metastases, and lumps in lymph nodes.
Lumakras’s mechanism of action
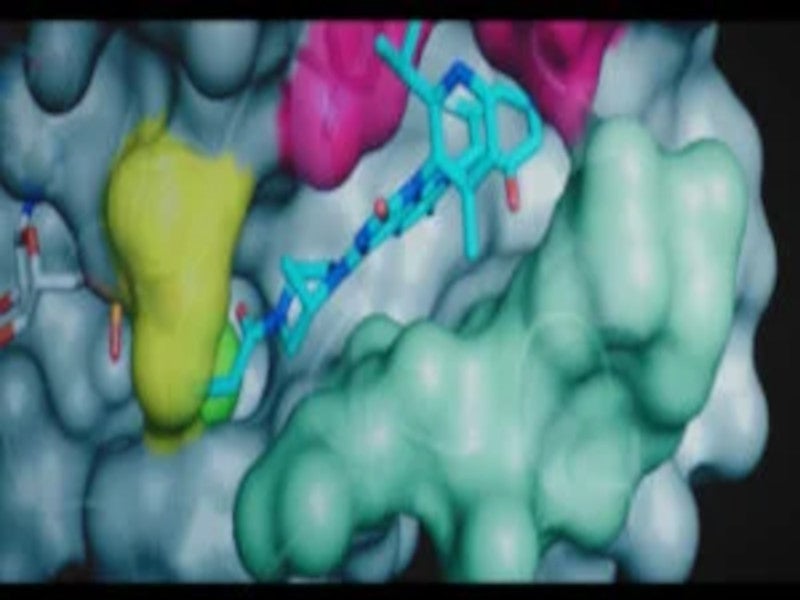
Sotorasib is a highly selective inhibitor of KRAS G12C that potentially suppresses the rapid proliferation of cancer cells.
The drug forms an irreversible, covalent bond with the cysteine residue of KRAS G12C, holding the protein in its inactive form that blocks oncogenic signalling. This results in apoptosis only in KRAS G12C tumour cells.
Clinical trials on Lumakras
The FDA’s accelerated approval of Lumakras was based on results from CodeBreaK 100, a single-arm, open-label, multi-centre clinical trial. The study enrolled a subset of patients with locally advanced or metastatic NSCLC with KRAS G12C mutations.
CodeBreaK 100 was the largest clinical trial conducted exclusively on the KRAS G12C-mutated NSCLC patients. A total of 126 patients were included in the study, of which 124 were confirmed with KRAS G12C-mutated NSCLC.
The study’s primary endpoint was the overall response rate (ORR) and duration of response (DOR), as assessed by a blinded independent central review (BICR) according to version 1.1 of the Response Evaluation Criteria in Solid Tumours (RECIST) specification.
All patients received at least one prior line of systemic therapy for metastatic NSCLC. In the study, patients received 960mg of sotorasib once a day until disease progression or unacceptable toxicity.
Sotorasib achieved a 36% ORR with a median response duration of ten months. The disease control rate, including patients achieving complete and partial responses or stable disease for more than three months, was 81%.
The most common side effects reported in Lumakras-treated patients in the clinical trial included diarrhoea, musculoskeletal pain, nausea, fatigue, liver damage and coughs.
The results from the Phase Ⅰb CodeBreaK 101 study for evaluating Lumakras’s safety and effectiveness in treating patients with KRAS G12C-mutated cancers were announced in October 2021.
The trial comprised two combinations, namely Lumakras with afatinib and Lumakras with trametinib. It enrolled 33 patients with KRAS G12C-mutated NSCLC for the Lumakras-afatinib combination arm, including five who previously received the Lumakras monotherapy.
The first cohort (cohort I) included ten patients who were administered 20mg of afatinib and 960mg of sotorasib, whereas the second cohort (cohort II) included 23 patients who were given 30mg of afatinib and 960mg of sotorasib.
The ORRs in cohorts I and II were 20% and 35% respectively, while the disease control rate in the two cohorts was 70% and 74% respectively.
The results also showed that heavily pre-treated patients with KRAS G12C-mutated solid tumours, including those who previously received KRASG12C inhibitor treatment, had anti-tumour activity when they were given a combination of Lumakras and trametinib.
A total of 41 patients volunteered for the Lumakras-trametinib combination arm, including 18 patients with NSCLC, 18 with colorectal cancer (CRC), and five with other solid tumours.
The partial response rate in patients with CRC with no prior KRASG12C inhibitor experience was approximately 9% while the disease control rate was 82%. The partial response rate in patients with prior KRASG12C inhibitor treatment was 14%, while the disease control rate was 86%.
The results indicated a partial response rate of 20% and a disease control rate of 87% in patients with NSCLC who did not previously receive KRASG12C inhibitor treatment. The disease control rate in patients with prior KRASG12C inhibitor treatment was 67%.
The most common adverse reactions were elevated levels of alanine and aspartate aminotransferases.