The product of a joint development programme between Swiss companies Speedel and Novartis, Rasilez (aliskiren) is an oral renin inhibitor indicated for the treatment of hypertension. The first orally administered drug in this new class of antihypertensive agents, Rasilez (aliskiren) has been evaluated in over 8,000 patients with high blood pressure.
Following successful completion of pivotal phase III trials, Novartis filed for regulatory approval in both the US and Europe, securing marketing authorisation in March and August 2007. In the US, the drug is marketed under the brand name Tekturna.
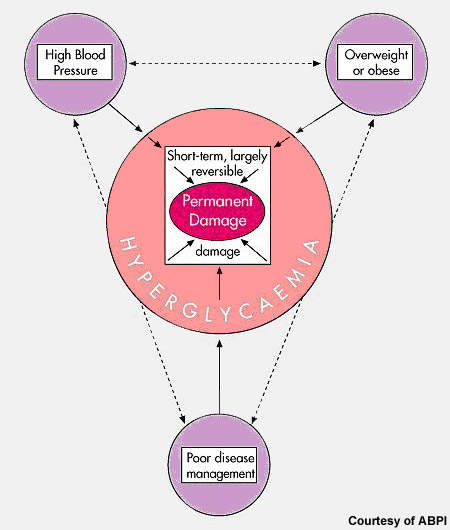
Hypertension and increased cardiovascular risk
Increased risk of cardiovascular morbidity and mortality is positively, continuously and independently associated with rising blood pressure, making optimal control of blood pressure an important goal in people with elevated blood pressure.
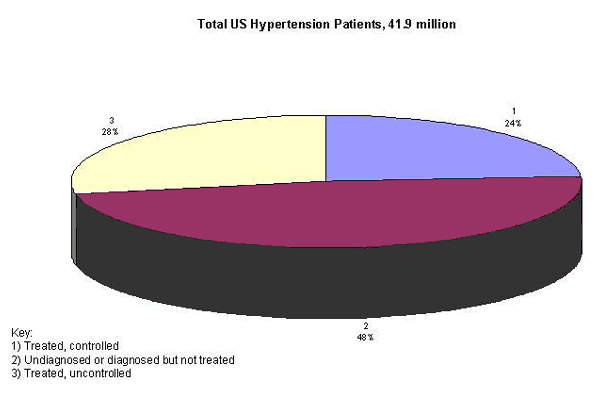
Although several classes of drugs exist to treat hypertension, many patients receive sub-optimal therapy. Estimates suggest that anywhere between 40% and 70% of patients fail to reach target blood pressure levels with currently available treatments.
There is therefore still considerable unmet clinical need in hypertension.
New approaches to antihypertensive therapy may help bridge current treatment gaps and enable more patients to reach blood pressure targets. Renin inhibitors represent the first of a new generation of antihypertensives with potential to improve the management of hypertension.
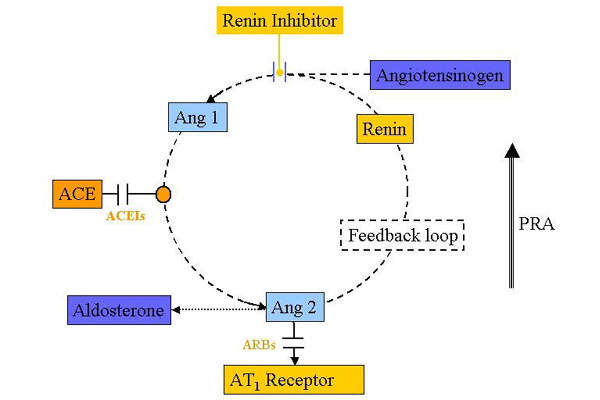
Targeting the renin angiotensin system (RAS)
The RAS is one of the key regulators of blood pressure, in which the enzyme renin plays a pivotal role. Renin, a proteolytic enzyme produced in the kidney, converts angiotensinogen in the blood to angiotensin-I.
This in turn is converted by angiotensin converting enzyme (ACE) to angiotensin II. Angiotensin II then stimulates the adrenal cortex to produce aldosterone, which is responsible for reabsorption of sodium and water from the urine in exchange for potassium and hydrogenions.
Drugs that target the RAS are already available as potent antihypertensive agents: the ACE inhibitors and angiotensin II receptor antagonists (ARBs) or sartans as they are commonly known. These classes of antihypertensive drugs block RAS “down stream” of renin.
Researchers believe that inhibition of renin may have important benefits over other strategies that target RAS. Renin inhibitors lower plasma renin activity (PRA) and may as a result have potential for improved end-organ (heart and kidney) protection.
PRA is a surrogate marker for diseases, such as myocardial infarction and chronic renal disease.
Rasilez demonstrates blood-pressure lowering effects
In initial clinical trials Rasilez (aliskiren) demonstrated potent dose-dependent blood pressure reduction in patients with hypertension. Subsequent large-scale, phase III trials have confirmed these early findings and provided evidence that Rasilez (aliskiren):
- Produces double digit reductions in systolic and diastolic blood pressure when administered as monotherapy accompanied by placebo-like tolerability
- Achieves 24-hour blood pressure control, notably in the early morning when there is often a surge in blood pressure
- Produces no rebound effect upon withdrawal; important if a patient forgets to take medication
- Superior blood pressure lowering when co-administered with ACE inhibitors, ARBs, diuretics and calcium channel blockers
- Well tolerated when co-administered with other antihypertensive drugs
- Appears safe and well tolerated in long-term use (> 12 months)
- Has potential for improved end-organ protection
Marketing commentary
It is estimated that over a billion adults in the world have hypertension and that by 2025 this will have risen to over 1.5 billion. Given its global prevalence, the market for antihypertensive drugs is large and growing.
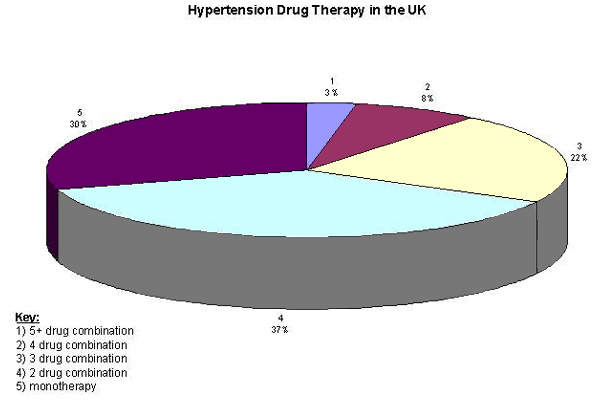
Recognition that many patients with hypertension need two or more drugs to achieve optimal blood pressure control has been a major driver of growth in this sector of the CVD market. Drugs with potential to further improve blood pressure control and prevent end-organ damage will find a ready market.
Oral renin inhibitors, of which Rasilez (aliskiren) is the most advanced, represent the first new treatment for hypertension since the introduction of the ARBs just over a decade ago.