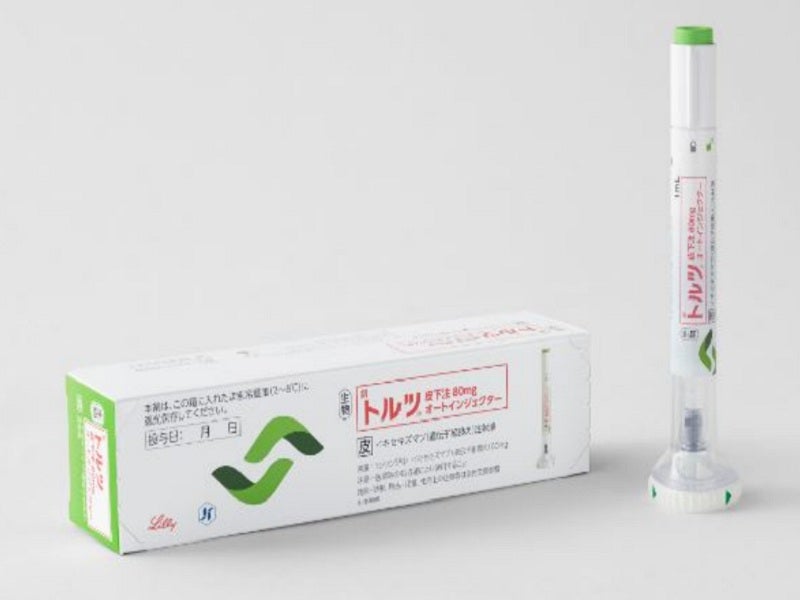
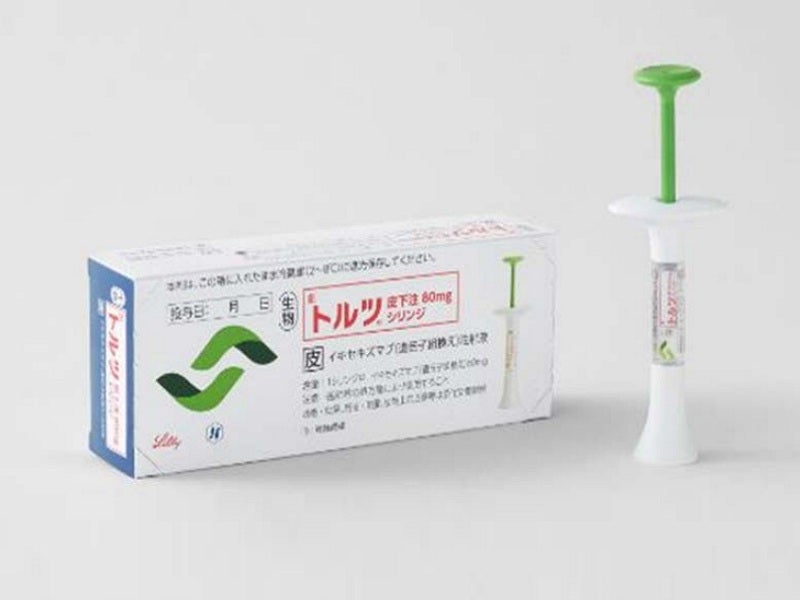
Taltz® (ixekizumab) is the first humanised interleukin (IL)-17A antagonist therapy approved for the treatment of active non-radiographic axial spondyloarthritis (nr-axSpA) in adult patients with objective inflammation signs.
The therapy is now eligible to treat all types of axSpA including ankylosing spondylitis (AS), or radiographic axSpA, and nr-axSpA.
It is a sterile, preservative-free, clear and colourless to a slightly yellowish solution available as auto-injector and prefilled syringe for subcutaneous administration. Both the injections contain a single dose of 80mg/ml solution of Taltz.
Taltz approvals
Taltz received its initial Food and Drug Administration (FDA) approval for the treatment of moderate to severe plaque psoriasis in patients aged six years and older in 2016. It is also approved for the treatment of radiographic axSpA and psoriatic arthritis in the US.
Eli Lilly launched the drug in Japan with Torii Pharmaceutical for the treatment of psoriasis vulgaris, arthropathic psoriasis, pustular psoriasis and psoriatic erythroderma in November 2016.
The company submitted a supplemental biologics license application (sBLA) for Taltz to the US FDA to treat adult patients with nr-axSpA, in July 2019. The drug received approval in June 2020.
Taltz is available in Europe for the treatment of plaque psoriasis and psoriatic arthritis. The European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion for the marketing authorisation of the drug to treat radiographic axSpA, and nr-axSpA, in April 2020.
In Canada, Taltz is approved for radiographic axSpA, plaque psoriasis, and psoriatic arthritis.
Axial spondyloarthritis signs and symptoms
Axial spondyloarthritis is a chronic inflammatory disease that primarily affects the sacroiliac joints and the spine, causing chronic back pain and fatigue due to inflammation. The disease includes both the radiographic and non-radiographic axSpA.
Approximately 2.3 million people in the US live with axSpA, of which 50% accounts to nr-axSpA. The disease is characterised by the damage in the sacroiliac joints structure, recognised on the X-ray for patients with radiographic axSpA. Non-radiographic axSpA patients do not clearly demonstrate the structural damages radiographically, which often leaves them undiagnosed.
Worsening of the disease causes more harm to the joints and spine and leads to a more serious condition called ankylosing spondylitis. Approximately 30% of the nr-axSpA patients get the ankylosing spondylitis within two to 30 years.
The most common symptom of the disease is back pain with other symptoms including weakness, joint stiffness or swelling, swollen fingers and pain in heels.
Taltz’s mechanism of action
Ixekizumab is a humanised IgG4 monoclonal antibody produced in a recombinant mammalian cell line by recombinant DNA technology. It binds to IL-17A cytokine selectively and blocks its interaction with the IL-17 receptor, a natural cytokine in the body responsible for inflammatory and immune responses. This inhibits the release of those cytokines and chemokines in the body which alerts the immune cells.
Clinical studies on Taltz
FDA approval of Taltz for the treatment of nr-axSpA is based on the results of a 52-week, multi-centre, randomised, double-blind, placebo-controlled phase III clinical study, COAST-X.
The study evaluated the safety and efficiency of the drug with the primary goal of improvement in signs and symptoms of nr-axSpA in patients, under the Assessment of SpondyloArthritis International Society 40 (ASAS40) response.
In the study, 304 patients were randomised to receive either 80mg Taltz every four weeks or two weeks or placebo. At 16 weeks, 35% of patients who received Taltz every four weeks and 40% of patients receiving Taltz at every two weeks achieved ASAS40 response compared to only 19% of patients receiving placebo.
At 52 weeks, 30% of patients receiving Taltz every four weeks and 31% of patients receiving Taltz every two weeks achieved ASAS40 response compared to 13% of patients treated with placebo.
The safety profile of the drug was found to be consistent with the previously reported data for other approved conditions.
Some common side effects observed in the patients during the clinical trials are injection site reactions, infections in the upper respiratory tract, nausea and fungal infection.