Xeljanz (tofacitinib) is a janus kinase (JAK) 3 inhibitor developed by Pfizer as a treatment for the symptoms of rheumatoid arthritis.
The drug has been studied for the treatment of a number of other diseases, including psoriasis and inflammatory bowel disease.
Almost 5,000 patients were administered the drug as part of the clinical trials, which were held in more than 350 centres in 35 countries. Four deaths have been reported during trials in 2011. Pfizer has announced that one of those deaths occurred as a result of tofacitinib, but the other three were not associated with the drug.
These deaths and the side effects of the drug delayed the anticipated approval of the drug. In November 2012, Xeljanz was approved by the US Food and Drug Administration (FDA) for treating rheumatoid arthritis.
Rheumatoid arthritis
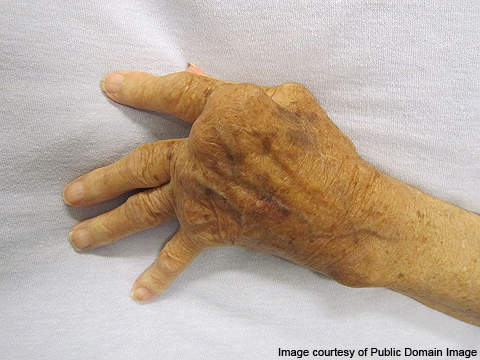
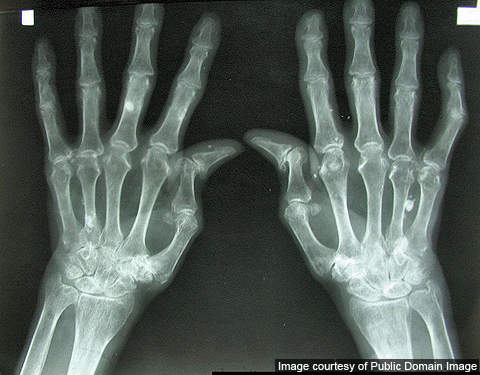
Rheumatoid arthritis is a chronic illness that affects the joints, surrounding tissues and other organs. The severity of the condition varies from person to person.
The symptoms at the initial stages include fever, fatigue, weakness, loss of appetite and swelling. This is followed by joint pains, which usually occur on both sides of the body.
Other symptoms include chest pain when breathing, numbness and burning sensation in eyes, hands and feet. Destruction of joints usually occurs one or two years after the appearance of first symptoms.
Xeljanz mechanism of action
Xeljanz contains JAKs, which transfer signals from cytokine to regulate immune cell function. Within the signalling pathway, JAKs also activate phosphorylate and Signal Transducers and Activators of Transcription (STATs) which regulate intracellular activity.
Xeljanz works by inhibiting the activation of phosphorylate and STATs. The drug is available in 5ml dose which is administered twice daily.
Tofacitinib clinical trials
Data released from the pre-clinical and clinical trials has shown that tofacitinib has the ability to relieve the symptoms of autoimmune diseases such as rheumatoid arthritis.
Phase I trials conducted to compare the bioavailability of oral and intravenous tofacitinib were completed by August 2010.
Phase II trials were conducted on Japanese rheumatoid arthritis patients who were previously medicated and showed low response with methotrexate. Around 140 people were enrolled at various centres and were given 1mg, 3mg, 5mg or 10mg dose of tofacitinib or a placebo. Reduced rheumatoid arthritis activity was observed in more than 72% of the patients. Safety and efficacy of the drug was also observed and no deaths occurred.
Six Phase III trials were conducted in patients with moderate-to-severe Rheumatoid Arthritis. The trials included ORAL Start, ORAL Sync, ORAL Scan, ORAL Solo, ORAL Step and ORAL Standard.
The results of the first showed that around 60% of the patients who were given either a 5mg or 10mg dose twice daily reached ACR20 – a 20% reduction in the number of swollen and tender joints.
ACR 50 was reached by around 31.1% and 36.8% of these patients respectively, while ACR70 was reached by 15.4% and 20.3%. The percentage of patients who did not even reach 20% improvement was 34%.
Better improvement was observed in patients being administered with tofacitinib compared to those given the placebo.
The results of the second Phase III trial (ORAL Sync) were released in March 2011. Around 792 patients with moderate-to-severe rheumatoid arthritis were enrolled and administered either 5mg or 10mg twice daily or a placebo. Tofacitinib was superior to the placebo in terms of reducing the signs and symptoms of rheumatoid arthritis. The study met all the primary endpoints.
The third Phase III trial (ORAL Scan) was a two-year trial in which patients were administered either 5mg or 10mg of tofacitinib twice daily, or a placebo, in combination with Methotrexate.
First year data was released in April 2011, revealing that the 5mg patient group showed increased improvements in the ACR20 response rates at six months when compared to the placebo. The safety and efficacy was consistent. The primary endpoint was met for the 10mg twice daily group. The ORAL Standard trial was a 12-month study and showed low response when patients were administered methotrexate. Hence, the patients were randomised to receive either tofacitinib or adalimubab, with stable background of Methotrexate.
In the ORAL Step trial patients were administered either 5mg or 10mg twice daily. The safety and efficacy data was found to be consistent and the study met its primary endpoint.
In November 2012, Pfizer agreed with FDA to initiate post-marketing clinical trials on Xeljanz to study its long-term effects on heart disease, cancer and serious infections.
Marketing Xeljanz in the US
Related project
Abatacept (Orencia) – Treatment for Rheumatoid Arthritis (RA)
Abatacept (Orencia) was developed by Bristol-Myers-Squibb for the treatment of rheumatoid arthritis (RA). It is a biologic drug which contains a selective co-stimulation modulator.
Xeljanz is the first JAK inhibitor to be approved in the last decade. It is expected to be a huge success in the market. The sales are expected to be $346m in 2013 and $1.65bn by 2016.
The drug faces competition from other approved drugs including Abbot Laboratories’ Humira, Bristol-Myers Squibb’s Abatacept and Janssen Biotech’s Remicade.