Xgeva (denosumab) is a preventive medicine developed by Amgen to check the spread of cancer to the bones. It was approved by US Food and Drug Administration (FDA) as the first and only rank ligand inhibitor for the prevention of skeletal-related events (SREs) in patients with bone metastases from solid tumours in 2010.
Xgeva is not indicated for the prevention of SREs in patients with multiple myeloma.
The drug is also approved by the European Medicine Agency (EMA) and the FDA for the treatment of osteoporosis in post-menopausal women. The FDA additionally approved the drug for its application in preventing the spread of cancer to the bones.
Xgeva also received approval in Canada for minimising the risk of developing SREs in patients with bone metastasis from breast cancer, prostate cancer, non-small cell lung cancer and other solid tumours.
Amgen received marketing authorisation for the drug in July 2011 for the prevention of SREs in adults with bone metastases from solid tumours. Approval applies to all 27 EU states with an additional one year of marketing exclusivity.
In June 2013, the FDA approved Xgeva for the treatment of adults and skeletally mature adolescents with giant cell tumour of bone (GCTB) that is unresectable or where surgical resection is likely to result in severe morbidity. The FDA also granted Xgeva orphan drug designation for this disease.
Xgeva was approved for the treatment of hypercalcemia of malignancy (HCM) refractory to bisphosphonate therapy in December 2014.
GSK will market the drug in the countries where Amgen does not have a presence.
Bone metastasis
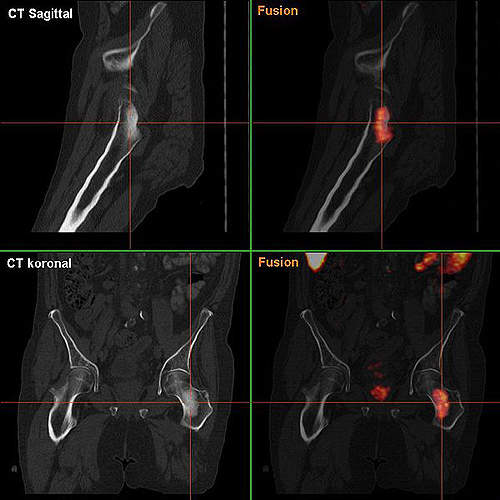
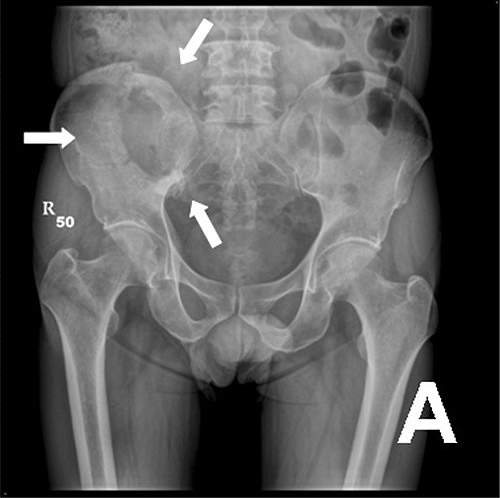
When a person develops cancer, there is a high risk that it spreads to other parts of the body. This process is called metastasis. It occurs in more than 1.5 million people suffering from cancer. Several types of cancers, including lung, breast, prostrate, kidney, thyroid and multiple myeloma’s, can lead to bone metastasis.
Hypercalcemia of malignancy (HCM)
HCM affects patients with advanced cancer, including those with haematological malignancies. It occurs most often in patients with squamous cell cancer, breast cancer, kidney cancer, myeloma and lymphoma.
Mechanism of action
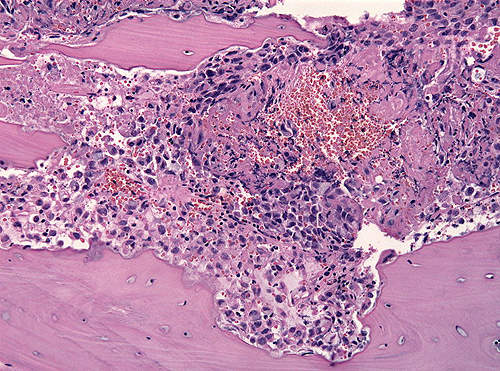
The bone tissue damage is caused by cells called osteoclasts which remove the matrix. These bone cells (osteoclasts) are activated by a protein called RANK ligand (RANKL).
A monoclonal antibody, Xgeva binds to RANKL and hinders it from activating osteoclasts. The inhibition decreases the differentiation, activity and survival of the osteoclastic bone cells. The drug reduces tissue damage and avoids bone erosion and breakage.
Overdose of the drug can lead to severe reduction of calcium in blood and osteonecrosis of the jaw (ONJ), a disease which occurs due to reduced flow of blood.
Clinical trials
Three clinical trials were conducted to discover the effectiveness of Xgeva. Phase I clinical trials were conducted in 2008 in Japan to study the safety, pharmacodynamics and pharmacokinetics of denosumab in women suffering from breast cancer.
Phase II clinical trials were conducted in 2009 to evaluate the drug’s effectiveness in the prevention of relapsed multiple myeloma. Phase III trials were conducted to evaluate the prevention of SREs in patients suffering from breast and prostrate cancer.
The Phase III trial included a randomised, double-blind study to compare the delaying time of first-on study SREs of Xgeva and Zometa in advanced cancer patients. The study also compared the effectiveness of Xgeva and Zometa in the prevention of SREs in patients suffering from advanced breast cancer and prostate cancer.
The first comparative study enrolled 1,776 cancer patients with solid tumours and bone metastasis. Patients were administered either 120mg of denosumab or 4mg of Zometa every four weeks. Denosumab, with median time of 19 months, was superior to Zometa with 14.4 months of median time to first on-study SRE.
For the second study, 2,046 breast cancer patients were enrolled. Two groups of patients were formed and administered either 120mg of denosumab or 4mg of Zometa in a 15 minute infusion. The doses were given once in four weeks. Results of the study showed that Xgeva was more effective than Zometa in delaying the time of SREs in patients suffering from advanced breast cancer.
For the third study, 1,900 men with advanced prostate cancer were enrolled. Results showed that Xgeva was superior to Zometa in delaying the time and the primary endpoint was met.