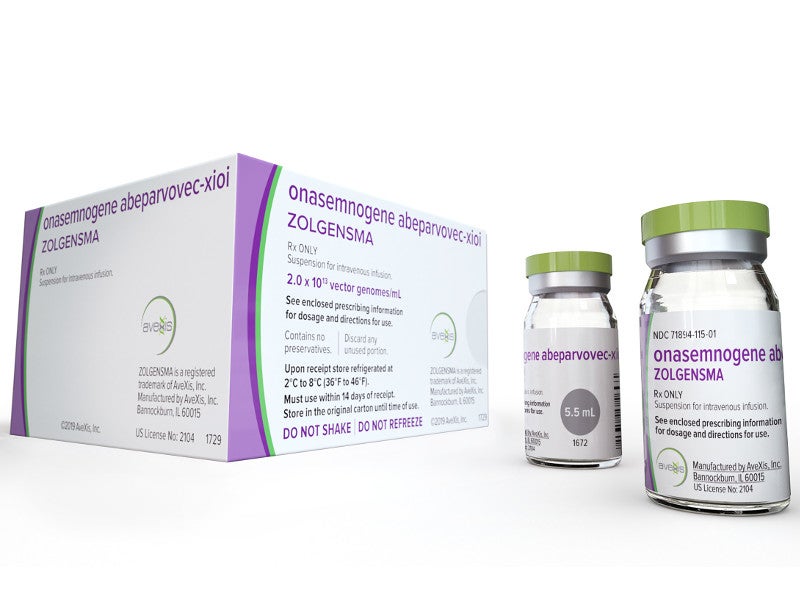
Zolgensma® (onasemnogene abeparvovec-xioi) is an adeno-associated virus vector-based prescription gene therapy used to treat paediatric patients with spinal muscular atrophy (SMA).
Originally developed by AveXis, the drug became a part of Novartis’ portfolio after it acquired AveXis in May 2018 and renamed Novartis Gene Therapies.
Priced at $2.1m for five years, the drug is one of the most expensive drugs to be approved. AveXis’ OneGene Program™ will provide personalised support to the patients’ families, including treatment information and financial assistance during the treatment period.
In August 2022, Novartis announced its plans to update the labelling of the drug to specify that fatal acute liver failure was reported due to the use of the drug. The announcement was made after two patient fatalities from acute liver failure were reported.
Regulatory approvals
Novartis submitted a biologics license application (BLA) for the drug to the US Food and Drug Administration (FDA) in September 2018, which was granted priority review status in December 2018. The FDA approved the drug in May 2019.
The therapy holds FDA breakthrough therapy and European priority medicines (PRIME) designations. It was reviewed under an accelerated assessment procedure by the European Medicines Agency (EMA) and granted Conditional Marketing Authorisation in 2020.
The Japanese Ministry of Health, Labour and Welfare (MHLW) provided SAKIGAKE designation to Zolgensma® and approved the drug in March 2020 to treat SMA type 1 in patients aged below two years, including those who are pre-symptomatic at diagnosis.
Zolgensma® was approved by Health Canada in December 2020 for the treatment of SMA.
Spinal muscular atrophy causes and symptoms
SMA is a rare genetic disease that deteriorates the neuromuscular functioning of the body by causing motor neuron loss and associated muscle weakness and paralysis.
The disease is caused by a genetic defect in the survival motor neuron (SMN) gene, which encodes the SMN protein essential for the survival of motor neurons. An estimated one in 10,000 babies is born with the disease.
SMA is categorised into SMA type 1 and SMA type 2. The first form is a more severe variant, causing rapid motor neuron loss and resulting in death or permanent ventilation support requirement in more than 90% of patients if left untreated. SMA type 2 leads to mortality in more than 30% of patients by 25 years of age.
Some common symptoms of SMA are muscle loss and weakening in infants after birth and difficulty in feeding and breathing.
Mechanism of action
Zolgensma® is an adeno-associated virus (AAV) 9-based gene therapy designed to deliver a copy of the SMN1 gene to encode for human SMN protein.
It is a recombinant form of self-complementary AAV9, which contains human SMN protein-encoding transgene.
The Zolgensma® drug is available in single-use vials, each containing a nominal concentration of 2.0×1013 vector genomes (vg) per millilitre for intravenous infusion.
Clinical studies on Zolgensma
FDA approval of the drug was based on the positive outcomes of two open-label, non-randomised, single-group, single-dose clinical studies named STR1VE and START. STR1VE was a Phase III clinical trial enrolling 21 patients with infant-onset SMA, while START is a Phase I clinical trial.
START, a 24-month clinical study, was conducted to evaluate the safety and efficacy of the drug in 15 patients. Three subjects enrolled in a low-dose cohort, while the remaining three enrolled in a high-dose group. The primary endpoint of the study was survival without permanent ventilation.
At the end of the 24 months, 12 patients who received a high dose no longer required permanent ventilation, representing event-free survival. During the trial, approximately 91.7% of the evaluated patients were able to hold their heads erect, while 83.3% achieved the ability to sit without support for more than ten seconds and 75% for more than 30 seconds. Furthermore, 16.7% of patients were able to stand alone and walk with assistance.
Event-free survival and support-free sitting of patients for at least 30 seconds were the primary endpoints of the 24-month STR1VE trial. Out of the 21 patients, 19 no longer needed permanent ventilation and 47.6% were able to sit without support for at least 30 seconds.
The most common adverse events (AE) reported in the patients were increased aminotransferases and vomiting.
AveXis also reported positive interim data from the Phase I/II STRONG trial, assessing the intrathecal formulation of the drug in SMA patients aged two to five years.
In October 2019, the FDA placed a partial clinical hold on studies of intrathecal Zolgensma®, after AveXis reported safety events in a pre-clinical study.
During animal tests, Zolgensma® was observed to be associated with dorsal root ganglia (DRG) mononuclear cell inflammation and neuronal cell body degeneration or loss in some cases.
The FDA also received a report from AveXis in August 2019, highlighting a manipulation issue with some data obtained from animal testing. The US regulator is assessing the data while recommending the continued market availability of the drug.
The partial clinical hold was lifted by the FDA in August 2021 based on a non-clinical toxicology study conducted in non-human primates (NHP).
The approval by Japan’s MHLW for the drug was based on the Phase Ⅰ START, START LTFU, and Phase III SPR1NT (intrathecal injection) studies.
The Phase III SPR1NT study was an open-label single-arm trial conducted to evaluate the safety and efficacy of one time IV infusion of Zolgensma® in 15 pre-symptomatic patients with two or three copies of SMN2 and who are less than six weeks old.
The results of the SPR1NT study showed that 100% of the patients met the primary endpoint of standing without assistance for more than three seconds by 24 months of age and 93% walked independently.
Ongoing clinical trials
START LTFU (Long-Term Follow-Up) is an ongoing, observational, long-term follow-up study of patients who showed symptoms of SMA and completed the START Phase I trial. The patients electively enrolled in the study and were divided into two cohorts.
As of December 2021, all ten patients in LTFU from cohort 2 were alive and off permanent ventilation. During the follow-up period, no previously accomplished motor milestones were lost. Two patients achieved a new milestone of standing with help.
Novartis initiated the SMART Phase III b global trial in 2021 in children with symptomatic SMA weighing up to 21kg to study the safety, tolerability, and efficacy of a one-time IV infusion of
Zolgensma®. The study is expected to complete in August 2023. The additional clinical data from the trial will be used to enhance the usage of the therapy in the EU and Canada.