Zyprexa (olanzapine) belongs to a newer group of antipsychotic medications called atypical antipsychotics and is widely used in the US. It is used mainly for the treatment of schizophrenia, psychotic disorders and bipolar disorders.
Pharmaceutical giant Eli Lilly manufactures Zyprexa, which is effective in treating overall symptoms of schizophrenia. Oral Zyprexa is prescribed for acute and maintenance treatment of schizophrenia in adults.
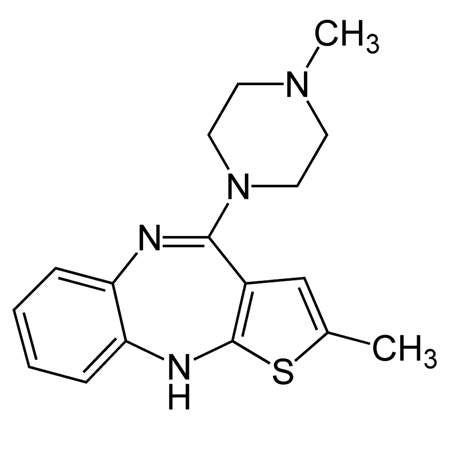
Zyprexa is part of the diazepines, oxazepines and thiazepines pharmacotherapeutic group. As a psychotropic agent Zyprexa belongs to the thienobenzodiazepine class. The drug has a higher affinity for 5-HT2 serotonin receptors than D2 dopamine receptors.
Zyprexa is the best selling drug in Eli Lilly’s 135-year history. Sales amounted to about $5bn in 2010, accounting for more than 20% of teh company’s total revenue.
Eli Lilly lost its patent for Zyprexa in October 2010, making the drug vulnerable to generic competition. However, industry observers believe that the company will be able to survive this competition because doctors may not be willing to switch to a cheaper version of the drug.
Eli Lilly also won approval for a long-acting form of the drug called Zyprexa Relprevv in December 2009. The patent for this version is valid for ten years.
With the new version, Eli Lilly is expected to make annual sales of $1bn by 2015.
The burden of schizophrenia and other mind-related disorders
Schizophrenia is a psychotic disorder belonging to a group of serious illnesses affecting the mind. It is among the top ten disabling conditions worldwide for young adults.
Patients with schizophrenia have poor functional outcomes and quality of life.
Adolescent psychosis poses a number of diagnostic and treatment challenges. Treatment costs for schizophrenia annually amount up to $60bn in the US.
Symptoms of bipolar disorder are treated with atypical antipsychotic medications. Zyprexa is used to treat severe or psychotic depression, including hallucinations or delusions experienced by patients.
Targeting psychosis and side effects
There are a number of different types of psychotic disorders, including schizophrenia, schizoaffective disorder, schizophreniform disorder, brief psychotic disorder, delusional disorder, shared psychotic disorder, substance-induced psychotic disorder, psychotic disorder due to a medical condition and paraphrenia.
Zyprexa is available in two formulations: as tablets, Zyprexa Zydis, which is an oral disintegrating tablet, and Zyprexa IntraMuscular for injection. The drug’s rate of affinity is quite low towards histamine, cholinergic muscarinic and alpha adrenergic receptors, similar to most atypical antipsychotics.
The mode of action is unknown but it is thought to involve antagonism of serotonin receptors. Extrapyramidal effects such as tardive dyskinesia and other therapeutic effects are the result of dopamine receptor antagonism.
Weight gain and sedation are caused by antagonising H1 histamine receptors. Other probable reasons for weight gain may be the antagonistic actions at 5-HT2C receptors. Zyprexa’s metabolism may be decreased or increased by agents that induce or inhibit CYP1A2 activity. Inducing agents include cigarette smoke and inhibiting agents may include the antibacterial ciprofloxacin.
Clinical trials lead to FDA approval
Zyprexa was approved by the US Food and Drug Administration (FDA) on 6 September 1996. On 24 December 2003, it was made a part of the Symbyax formulation. On 14 January 2004 it was approved for treating acute manic episodes and maintenance treatment of bipolar disorder.
The National Institute of Mental Health and Eli Lilly jointly funded a study called Prevention through Risk Identification, Management, and Education (PRIME). As part of the study, 60 patients with prodromal schizophrenia were treated with Zyprexa and results proved that it was effective but was associated with weight gain.
On 23 March 2009, it was made part of Symbyax for treatment-resistant depression.
Marketing commentary
Some adverse reactions have been identified with the use of Zyprexa. These include allergic reactions along with venous thromboembolic events and discontinuation reactions. Neuroleptic malignant syndrome, a potentially life-threatening reaction, has also been reported, raising further concerns over the use of Zyprexa.
Weight gain and an increased risk of diabetes are the most common yet also concerning side effects of Zyprexa. Lilly has announced that the feedback on weight gain, hyperglycaemia and hyperlipidaemia was included in the labels on Zyprexa and Symbyax. This was included after the FDA’s review of clinical data between August 2007 and July 2008.
However, press reports leaking documents on the risks and side effects of the drug have led to lawsuits against Lilly and Zyprexa’s future as a market-leading antipsychotic seems unsure.