
Growth hormone deficiency (GHD) causes developmental problems for children such as short stature, stunted bone development, and issues with fat regulation. Growth hormones (GH) can be administered, but the treatment duration in children is lengthy and longer-term effects have not been well studied.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
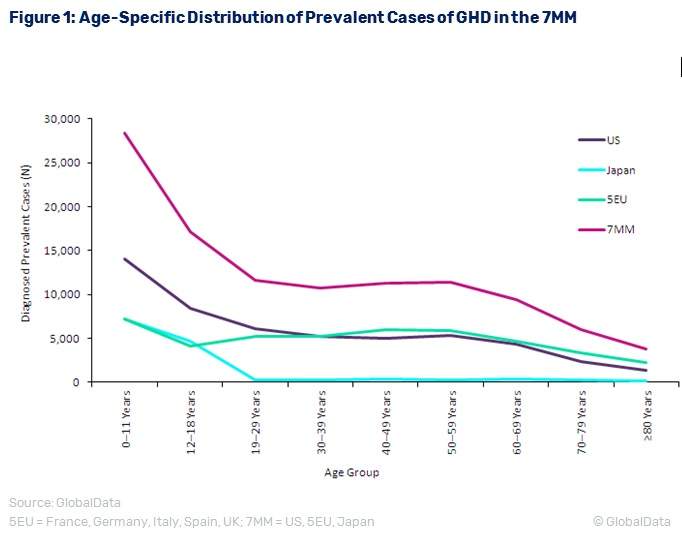
GlobalData’s analysis of GHD epidemiology in the seven major pharmaceutical markets (7MM) of the US, France, Germany, Italy, Spain, the UK, and Japan determined that the diagnosed prevalent cases of GHD for all ages will increase from 109,551 cases in 2016 to 112,450 cases in 2026 at an annual growth rate of 0.26%. Children aged less than 18 years old make up about 40% of the prevalent population, at 45,491 cases in 2016.
Figure 1 presents the age-specific trends of diagnosed prevalent cases of GHD in the 7MM. The treatment rate varies from 60%-82% in 2016, and is expected to increase in the next ten years. As the treatment period for GHD in children is often several months to years in duration, long-term effects of GH supplementation must be considered.
The long-term effects of GHD treatment in children are not well understood because the laboratory-based recombinant human GH drug has not been around for very long. Historically, the GH drug was scarce because it had to be isolated from human pituitary glands in cadavers. However, since the development of the artificial GH in the 1980s, GH has become much more readily accessible and more people have received the treated. It is only in the last few years that longitudinal data have become available in the first groups of GH-treated children since the 1980s.
One study conducted in France by Poldvin followed children who started GH treatment between 1985 and 1996. It found an increased risk of stroke in the treated population, with risks as much as 3.5 to seven times as high as children not treated with GH. Other studies of long-term effects of GH in children found no negative outcomes, but they had a shorter follow-up period and tended to focus on subgroups within the GHD-treated population.
One multicenter study conducted by Bakker examined GH treatment in Prader-Willi syndrome children with eight years of follow-up. This study did not find significant adverse effects on glucose homeostasis, serum lipids, blood pressure, or bone maturation.
The current GH treatment can be difficult to follow because it requires giving daily shots for many months / years. Many people may opt to not use it. However, as long-acting recombinant GH treatments that would only require one dose per week become available for use in the 7MM, more GHD patients may consider the treatment. More studies on the long-term effects of GH treatment would be necessary so parents and patients can weigh the potential benefits and risks associated with GH treatment.





