The swift development and deployment of messenger-RNA (mRNA) vaccines against the SARS-CoV-2 virus during the COVID-19 crisis has catapulted the pharmaceuticals industry into a new paradigm. RNA has the potential to underpin breakthrough treatments for a wide variety of diseases, including many cancers, and transform medicine as we know it.
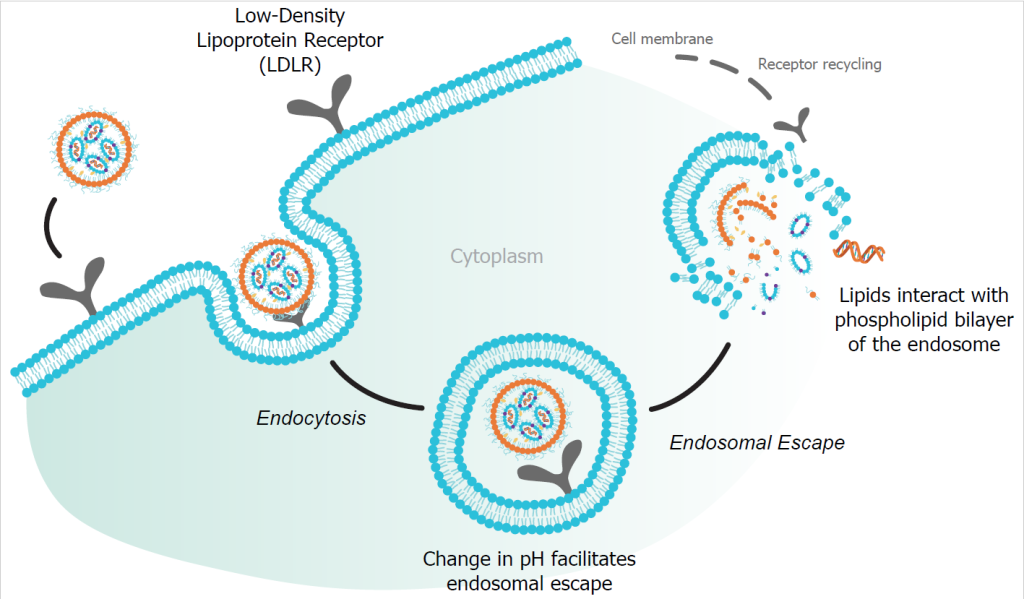
But the mRNA technology is not yet mature and there are no standardised manufacturing protocols yet. More challenges arise with the formulation of the mRNA delivery agent, like lipid nanoparticles (LNPs). Used in the COVID-19 vaccines, LNPs have shown a promising track record of protecting the mRNA cargo from degradation. They have proven to diminish cytotoxicity and organ toxicity in new and current treatments and improve the efficacy and pharmacokinetic profile of mRNA. This is due to two unique characteristics. Their modularity allows lipid excipients to be mixed, matched, and adapted to deliver a range of nucleic acid payload to different targets in the body. Their capacity, which facilitates the delivery of larger mRNAs and the co-delivery of multiple RNAs, enables new therapeutic mechanisms.
But Adam Crowe, Manager, Analytical Development at Precision NanoSystems (PNI) says that as the technologies facilitating LNP-RNA therapeutics are in their infancy, there is no clear line between proof-of-concept data to final product – something which he says can surprise small-to-medium size biotech companies looking to scale their projects.
“At the small scale, manufacturing between 1 ml and 10 ml is typically not problematic, but it’s not straightforward to replicate processes and technologies once you want to produce tens of litres of formulations,” says Dr Crowe, who manages a team that develop novel analytical assays related to LNPs and nanomaterials for drug delivery. “Existing compositions have proven effective, but a complex landscape of intellectual property (IP) and licensing agreements make accessing these compositions complex for new parties when attempting to get to the clinic.
“Companies that want to develop drugs using these new modalities must not only find new sources of material to bring to a CDMO, but they should also understand the many variables that could cause the project to fail.
“If just one parameter changes you could have a catastrophe – and there are at least 30 to 40 parameters to consider. Some of the variables include the chemical composition of the LNP, the manufacturing process and of course the RNA itself, whose design and production must elicit the intended therapeutic outcome.”
Avoiding lipid impurities
According to Dr Crowe, one of the emerging challenges to the potency of an LNP are N-oxide-based impurities, originating from the ionizable lipid, which can deactivate the mRNA.
“The current type of LNP we use has four lipid components and one of these is known as an Ionizable lipid. This is the species that changes its charge depending on pH. These have really enabled the new generation of LNPs, but the problem is that Ionizable lipids are inherently more reactive. From a chemistry perspective when you have a tertiary amine – a reactive species – on it and you can get a whole lot of weird chemistry occurring – one of which is the main impurities that’s been identified within these nanoparticles.
“They can react with RNA and inactivate the RNA itself.”
The other impurity challenge arises from the chemical synthesising of the Ionizable lipid and the PEG lipid components. They can’t be derived from animal products, but they are often synthesised in low purity – 60%-70% pure. That is not an issue in an academic lab but becomes a big problem when you want to manufacture a therapeutic because you need to know what the impurities are down to 1% or 0.1% weight abundance.
“That can kill a lot of projects unfortunately,” says Dr Crowe. “That’s a big area of analytics where we would use different High Performance Liquid Chromatography (HPLC) approaches. Charged Aerosol Detection (CAD) is industry standard, but we also use liquid chromatography–mass spectrometry (LC-MS) approaches to identify what’s in the materials. The ability to produce high quality material, to understand its solubility and degradation behaviour and reactivity is something that we spend a lot of time doing as it’s generally not done in earlier phases of client work. This involves monitoring the material in different conditions. Does it degrade? Does it form new species? Does it react with the RNA or other components? This is increasingly becoming a challenge.” Particle size and polydispersity are also variables that can sink a project. Polydispersity is the range of particle sizes produced during manufacturing. LNPs are never produced at a single size, always a distribution, and the PDI measures how big that distribution is. If PDI gets large that means your manufacturing process has gone wrong because your particles are behaving differently, fusing, aggregating and not behaving as you want them to.
“Another area where we tend to see problems is in encapsulation efficiency,” says Andrew Kondratowicz, Bioassay Team Lead, Precision NanoSystems. “Where is your payload? Where is it allocated? Is it inside the particle’s intended location, stuck to the outside or even free in solution? The importance here is the whole point of the LNP is to protect the RNA and help deliver it. If it’s not inside, it really serves no purpose and there are a number of immunogenic and other complications if the RNA is outside. This is another example of what encapsulates at a small scale can fail to work at large scale.”
The importance of the ‘biological readout’
One of the big areas when developing these processes and optimising the composition, looking at all the different ways you can manufacture and produce it, is to have a ‘biological readout’. Crowe and Kondratowicz say you must be able to take your formulation and say: ‘does it have the intended effect I’m looking for?’.
“I think this is overlooked a lot, but we believe it is essential that the potency of the material is carefully monitored every time you make a small change,” explains Kondratowicz. “The bioassays are the canary in the coal mine of how well your process development is going. We can monitor hundreds of different variables in the LNP scaling process with bioassays and quickly flag it to our clients if something is going wrong. This is required from regulatory bodies too for filing your new drug product. Every formulation scales differently and so to ensure a programme’s success you need a bioassay in place.”
Crowe comments: “Scaling production of LNP-RNA sequences are just so fraught with things that can go wrong and so Andrew’s ability to screen at high capacity means that even if a batch fails – which is not uncommon given the complexity of these materials and how new the market is – PNI is able to help get our clients’ projects swiftly back on track for a successful outcome.
Kondratowicz adds: “In terms of ensuring the RNA quality itself, PNI can do formulation independent mechanisms of getting the RNA into the cell so that at least we go into the LNP formulation process knowing we are working with good RNA and avoiding a scenario where later on in the project something goes wrong and then realising it was the RNA. At least we take off that variable to help the project. We can produce in excess of 100 milligrams in-house but for larger batches, we can screen for the correct output and then tech transfer to another vendor who can replicate it in much larger scale.”
Outlook
The era of mRNA-based genomic medicines is on the horizon. The technology underpinning the COVID-19 vaccines is the tip of the iceberg. Next-generation mRNA vaccine designs that use self-amplifying RNA (saRNA) and circular RNA (circRNA) molecular formats are in development. With these, the RNA required could be up to 100 times less with increased efficacy and performance. The circRNA will not have the Poly-A tails and 5’ caps that cause so much of the quality problems in the manufacture of RNA. But the analytical technologies and processes required will get much more complex too and PNI is unique in its ability to analyse both of these already.
With so much uncertainty still surrounding the best approaches to process development and manufacturing of mRNA-LNP with the optimal economics, partnering with end-to-end solution providers like PNI who can provide evolving and tailored strategies to support fast-emerging industry needs will be key.
We will truly see the influence of mRNA technology in the future trajectory of vaccine development, oncology, and personalised medicine and PNI aims to support biotechnology leaders and academic research labs with access to an exclusive proprietary lipid library and expertise to develop a bespoke lipid nanoparticle formulation for genomic medicine.
PNI’s GenVoy Delivery Platform comprises off-the-shelf RUO reagents such as GenVoy-ILM™ and a library of proprietary lipids available as custom formulations. LNP manufacturing is scalable to advanced preclinical and clinical scales with NxGen™ NanoAssemblr® technology.


