Extracellular vesicles (EVs) are tiny, membrane-bound particles released by every cell type studied to date. Though small, they carry a biologically sophisticated cargo of proteins, lipids, and nucleic acids. Moving through the extracellular space, EVs function as specialised messengers delivering molecular signals that can recalibrate the behaviour of recipient and neighbouring cells (1-3).
Once EVs reach their destination they fuse with target cells and release their molecular cargo directly into the cells’ cytoplasm. At that moment, a decisive shift begins: the cell’s behaviour, phenotype, and overall function changes. This is the heart of why EVs have become a compelling focus in regenerative medicine. By delivering carefully packaged bioactive signals, EVs can nudge damaged tissues toward repair, help recalibrate an overactive or suppressed immune response, and encourage cells to proliferate. In many ways, EVs act like tiny biological messengers capable of reshaping a cell’s future from the inside out.
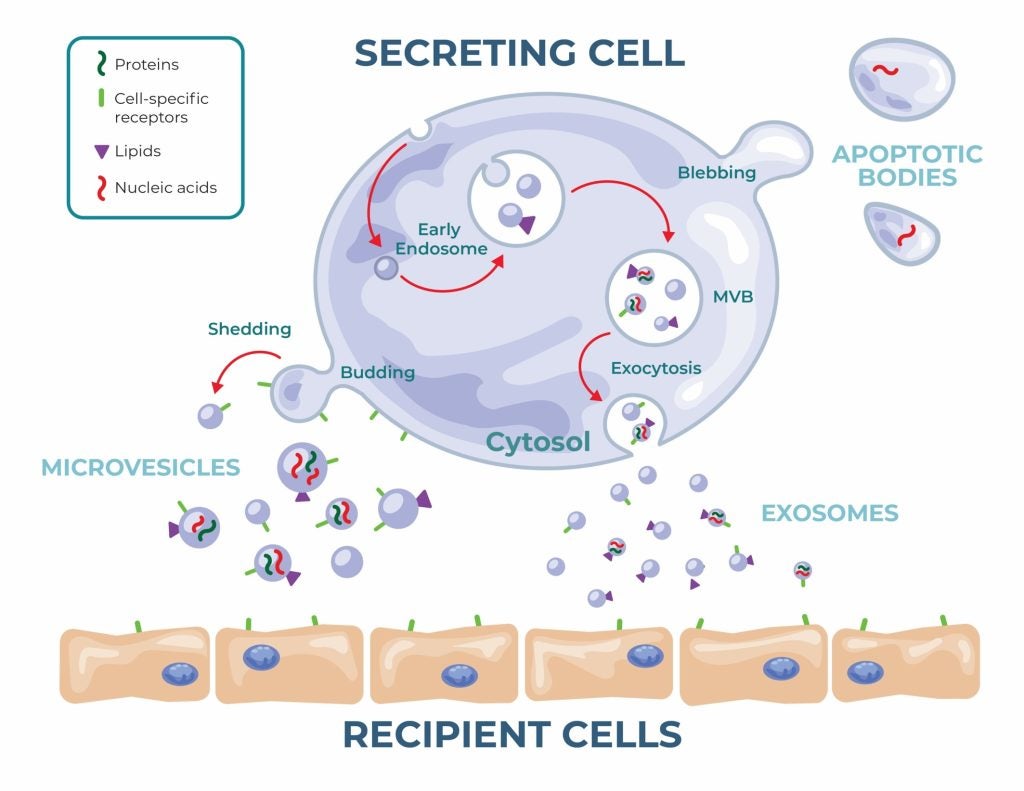
Although they are much smaller than cells, EVs have a similar envelope to the cell membrane and they can be harvested, even as the cells are being expanded. In regenerative medicine, they effectively represent a cell-free alternative to traditional cellular therapies.
EV’s advantages over cell therapies
While the science is in the early stages, several fundamental distinctions suggest that EVs could offer a safer, more effective alternative to cell-based therapies. An advantage of EVs over traditional cellular therapies is their ability to evade the immune system while homing in on specific cell types, including immune cells, cancer cells, and endothelial cells (4). The molecules and proteins on an EV’s membrane determine which cells it can recognise and interact with.
Unlike living cells, EVs cannot divide or generate new copies of themselves. This significantly reduces the risk of engraftment-related side effects, one of the major safety concerns associated with traditional cell therapies. According to published data, acute graft-versus-host disease occurs in approximately 28 to 52% of allogeneic cell transplant recipients (5).
When it comes to processing and storage, EVs offer additional advantages. Unlike cell therapies, they do not require cryopreservation media or liquid nitrogen, and there is also evidence that they retain biological activity after lyophilisation (6). This stability makes EVs easier to store, transport, and distribute (7).
Applications for EVs
There are three types of EVs – exosomes, microvesicles, and apoptotic bodies differentiated by their size, molecular composition, and release pathways (8). In the world of EVs, size is not just a descriptor; it often shapes their biological role, behaviour, and therapeutic potential.
While most of the research is currently produced in animal models, the science shows promise. EVs sourced from mesenchymal stem cells (MSCs) have already demonstrated the ability to reduce inflammation in pulmonary hypertension and osteoarthritis in mice (9-11).
In neurology, EVs are showing ability to cross the complex and challenging blood-brain barrier. In preclinical studies, Dong-ki et al. demonstrated that stem cell-derived exosomes can support recovery from traumatic brain injury in animal models (12). EVs have also shown potential at slowing the progression of amyotrophic lateral sclerosis through neuroprotective effects.
And that’s only half of the story. Beyond their naturally occurring therapeutic benefits, EVs can be engineered in the laboratory to carry a wide range of specific cargo, from small molecules to vaccines, nucleic acids, and enzymes. The concept is like the approach used in synthetic lipid nanoparticles (LNPs), one of the major technological successes behind COVID-19 vaccine development. However, EVs hold a key advantage: their natural biocompatibility and lower immunogenicity may result in fewer adverse reactions.
In one example, Haney et al. used extracellular vesicles to transport an active TPP1 enzyme to the brains of mice with Batten disease, resulting in reduced neurodegeneration and neuroinflammation (13). The nanocarrier approach is also showing potential in oncology, where EVs can be loaded with cytotoxic drugs and directed towards cancer cells, while their lipid membrane helps shield healthy cells from harmful off-target toxicity.
EV isolation challenges
Extracellular vesicles are a nascent but exciting area of drug development, combining natural regenerative potential with endless possibilities in drug delivery. However, EV manufacturing faces hurdles, particularly in achieving consistent, high-yield, and scalable manufacturing processes that meet the rigorous standards required for clinical translation.
Two manufacturing workflows are available depending on whether the EVs are isolated directly from biological fluids such as blood or from conditioned cell culture media. While the former is commonly used for biomarker development, the latter offers larger, more reproducible yields, offering clearer regulatory and characterisation pathways (14). From a manufacturing perspective, BBG Advanced Therapies (BBGAT), a full-service biomanufacturing organisation specialising in GMP-compliant manufacturing of cell-based and EV-based products, can scale media volumes up to 80 litres, then harvest and isolate the EVs from that material.
While collecting EVs from cell culture has improved scalability and helped pave a way for these therapeutics to enter clinical trials, several challenges remain. The inherent variability of cell-based applications influences production output, introducing potential for batch-to-batch inconsistency (15). Furthermore, scale-up for commercial production also poses another layer of complexity depending on the starting material source and the culture method used (16).
There are trade-offs to consider when choosing between primary cell lines and immortalised cell lines, as well as when working in 2D vs. 3D and dynamic culture systems. Each option offers distinct advantages but also introduces variability that can affect yield, purity, scalability, and cost. Compounding this is the historically limited characterisation of extracellular vesicle products, which adds another layer of difficulty to achieving consistent and reproducible manufacturing (17).
Overcoming consistency challenges
BBGAT offers a comprehensive suite of biomanufacturing services for companies looking to advance extracellular vesicle programs. The company has established a streamlined process designed to address many of the challenges inherent to EV production, helping reduce batch-to-batch variability and support consistently high-quality outputs.
The process begins with screening and selecting a single qualified donor whose cells establish a cell bank that can be assessed and qualified. Prior to manufacturing, BBGAT performs process development runs in which EVs are tested and characterised. Any necessary optimisation is completed at this stage, after which the finalised process is locked into the batch record to ensure consistency and reproducibility.
When it comes to the specifics of both upstream and downstream operations, the process is highly tailored to each client’s needs. The cell lines are derived from MSCs which can be extracted from a variety of sources and expanded from primary tissue or established cell vials. Upstream, cultures can be scaled in either a 2D, 3D or dynamic system formats using large volume systems such as Corning® CellSTACK®s or bioreactors to generate the required yields.
BBGAT utilises a tangential flow filtration system for EV enrichment during downstream purification. Because the filter is parallel to the flow, it can manage larger volumes and achieve higher recovery. It also introduces less shear stress and minimises damage to the product, which is critical for maintaining stability. This system also allows for a buffer exchange and concentration of the product if needed.
For the final steps, the team offers a range of fill-finish formulations and packaging formats, from vials to bags. Once the product is stored, their dedicated group of assay development experts design a comprehensive analytical panel to assess the EVs for stability and potency. This includes assessing concentration, size distribution, surface markers, total protein count, and specific content such as growth factors.
While a growing body of evidence supports the potential of EVs in regenerative medicine and targeted drug delivery, the field is still in its preliminary stages. As research progresses, the next five years is likely to bring more EV-based candidates into clinical evaluation, offering precise therapeutic delivery for a wide range of diseases. However, before that vision can be fully realised, biotech developers must establish large-scale, reproducible GMP manufacturing strategies that preserve the purity and structural integrity of these small particles. For many, the most effective path forward is partnering with advanced therapy specialists like BBG Advanced Therapies.
With special thanks to Samantha Dykes, PhD, Sr. Scientist, Process Development, BBG Advanced Therapies, Alia Mallah, PhD, Sr. Scientist, Process Development, BBG Advanced Therapies, Tyler Askelson, PhD, MS, Manager, Operations, BBG Advanced Therapies, Robert Reese, PhD Manager, Manufacturing Science & Technology, BBG Advanced Therapies, Joseph Higdon, MS, MBA, ASQ CPGP, Director Quality and Compliance for Advanced Therapies, BBG Advanced Therapies, Deidre Cacchillo, BSN, RN, Director of Marketing, BioBridge Global, Rogelio Zamilpa, PhD, Executive Director of Biomanufacturing, BBG Advanced Therapies.
To learn more about BBG Advanced Therapies, please download the whitepaper below.
References
(1) Buschow S. et al. (2009) MHC II in dendritic cells is targeted to lysosomes or T cell-induced exosomes via distinct multivesicular body pathways. Traffic. doi.org/10.1111/j.1600-0854.2009.00963.x (2) Melo SA et al. (2014) Cancer exosomes perform cell-independent microRNA biogenesis and promote tumorigenesis. Cancer Cell. doi: 10.1016/j.ccell.2014.09.005 (3) Baruah, J and Wary, K (2019) Exosomes in the Regulation of Vascular Endothelial Cell Regeneration. Frontiers in Cell and Developmental Biology. DOI=10.3389/fcell.2019.00353 (4) Phinney (2017) Concise Review: MSC-Derived Exosomes for Cell-Free Therapy. Stem Cells. doi: 10.1002/stem.2575 (5) DynaMed. Acute Graft-Versus-Host Disease. EBSCO Information Services. Accessed February 17, 2026. https://www.dynamed.com/condition/acute-graft-versus-host-disease-1 (6) Zhang (2020) Exosome: A Review Of Its Classification, Isolation Techniques, Storage, Diagnostic And Targeted Therapy Applications. Int. J. Nanomed. DOI: 10.2147/IJN.S264498 (7) Morgane E. Golan, ME and Stice, SL, (2024) Extracellular vesicle lyophilization for enhanced distribution to the point of care. Extracellular Vesicle. doi.org/10.1016/j.vesic.2024.100041 (8) Caruso, S., and Poon, I. K. H. (2018). Apoptotic cell-derived extracellular vesicles: more than just debris. Front. Immunol. 9:1486. doi: 10.3389/fimmu.2018.01486 (9) Lee, C., Mitsialis,; S Alex, Aslam, M., Vitali, S. H., Vergadi, E., Konstantinou, G., Sdrimas, K., Fernandez-Gonzalez, A., & Kourembanas, S. (2012). Vascular Medicine Exosomes Mediate the Cytoprotective Action of Mesenchymal Stromal Cells on Hypoxia-Induced Pulmonary Hypertension. https://doi.org/10.1161/CIRCULATIONAHA (10) Cosenza, S., Toupet, K., Maumus, M., Luz-Crawford, P., Blanc-Brude, O., Jorgensen, C., & Noël, D. (2018). Mesenchymal stem cells-derived exosomes are more immunosuppressive than microparticles in inflammatory arthritis. Theranostics, 8(5), 1399–1410. https://doi.org/10.7150/thno.21072 (11) Zhou, H., Shen, X., Yan, C., Xiong, W., Ma, Z., Tan, Z., Wang, J., Li, Y., Liu, J., Duan, A., & Liu, F. (2022). Extracellular vesicles derived from human umbilical cord mesenchymal stem cells alleviate osteoarthritis of the knee in mice model by interacting with METTL3 to reduce m6A of NLRP3 in macrophage. Stem Cell Research and Therapy, 13(1). https://doi.org/10.1186/s13287-022-03005-9 (12) Kim, D. K., Nishida, H., An, S. Y., Shetty, A. K., Bartosh, T. J., & Prockop, D. J. (2016). Chromatographically isolated CD63+CD81+ extracellular vesicles from mesenchymal stromal cells rescue cognitive impairments after TBI. Proceedings of the National Academy of Sciences of the United States of America, 113(1), 170–175. https://doi.org/10.1073/pnas.1522297113 (13) Haney, M. J., Klyachko, N. L., Harrison, E. B., Zhao, Y., Kabanov, A. V., & Batrakova, E. V. (2019). TPP1 Delivery to Lysosomes with Extracellular Vesicles and their Enhanced Brain Distribution in the Animal Model of Batten Disease. Advanced Healthcare Materials, 8(11). https://doi.org/10.1002/adhm.201801271 (14) Gardiner, C., Vizio, D. di, Sahoo, S., Théry, C., Witwer, K. W., Wauben, M., & Hill, A. F. (2016). Techniques used for the isolation and characterization of extracellular vesicles: Results of a worldwide survey. Journal of Extracellular Vesicles, 5(1). https://doi.org/10.3402/jev.v5.32945 (15) Huang, J., Chen, H., Li, N., Liu, P., Yang, J., & Zhao, Y. (2025). Emerging technologies towards extracellular vesicles large-scale production. Bioactive Materials, 52, 338-365. (16) Estes, S., Konstantinov, K., & Young, J. D. (2022). Manufactured extracellular vesicles as human therapeutics: challenges, advances, and opportunities. Current Opinion in Biotechnology, 77, 102776. doi: 10.1016/j.copbio.2022.102776 (17) Li, Z., Yan, J., Li, X., Chen, H., Lin, C., Zhang, Y., Gao, T., Zhang, Y., Shu, Y., Pan, S., Zhang, Y. (2025). Advancements in extracellular vesicles biomanufacturing: a comprehensive overview of large-scale production and clinical research. Frontiers in Bioengineering and Biotechnology, 13, 1487627. doi: 10.3389/fbioe.2025.1487627



