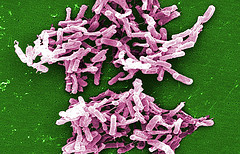
Sanofi Pasteur, the vaccines division of Sanofi, has begun Phase III clinical programme, known as Cdiffense, to assess an investigational Clostridium Difficile (C. diff) vaccine against a causative agent for life-threatening, healthcare-associated infections worldwide.
The company has designed the investigational vaccine to help protect at-risk individuals from C. diff, an emerging key causal factor of life-threatening, healthcare-associated infections (HAIs) globally.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Sanofi Pasteur research and development senior vice-president Dr John Shiver said CDI has become more frequent, severe and difficult to treat in recent years due to the emergence of difficult-to-manage strains of C. diff, raising control and prevent transmission concerns.
"Vaccination could be an efficacious, cost-effective and important public-health measure to protect individuals from C. Diff," Shiver said.
The safety, immunogenicity and efficacy profile of an investigational vaccine will be assessed in Cdiffense to help prevent primary symptomatic Clostridium difficile infection (CDI).
Recruitment of 15,000 adult volunteers at 200 sites across 17 countries has begun for a randomised, observer-blind, placebo-controlled, multi-centre, multi-national trial of Cdiffense Phase III clinical programme.
The study will include volunteers as old as 50 years and beyond, who are planning an upcoming hospitalisation or have had at least two hospital stays and received systemic antibiotics in the past year.
The risk of C. diff increases with age, antibiotic treatment and time spent in hospitals or nursing homes.
Image: Gram positive C. Difficile bacteria. Photo: courtesy of Janice Carr / CDC PHIL.





