According to GlobalData’s Clinical Trial Database, the number of overall decentralised clinical trials (DCT) has steadily increased from 250 trials in 2012, to 1,291 trials in 2021. There have been 950 trials in 2022 so far, and this is expected to increase to approximately 1,425 trials by the end of the year. From 2012 to 2016, both industry sponsors and non-industry sponsors had similar values regarding DCT usage. From 2016 onwards, more non-industry sponsors were using DCTs than industry sponsors.
The US dominates the landscape of DCTs from 2015 onwards, with a sharp increase occurring in 2021. The UK, Germany, Canada, and Australia have similar values for DCT usage. An increase in DCT usage also occurs for these nations from 2020 to 2021. The sharp increase between 2020 and 2021 could be attributed to the Covid-19 pandemic. Additionally, the top five nations are all high-income nations. High-income countries are far more likely to conduct clinical trials with virtual components than middle/lower income countries, as high-income countries have the regulatory support required to adopt these new approaches.
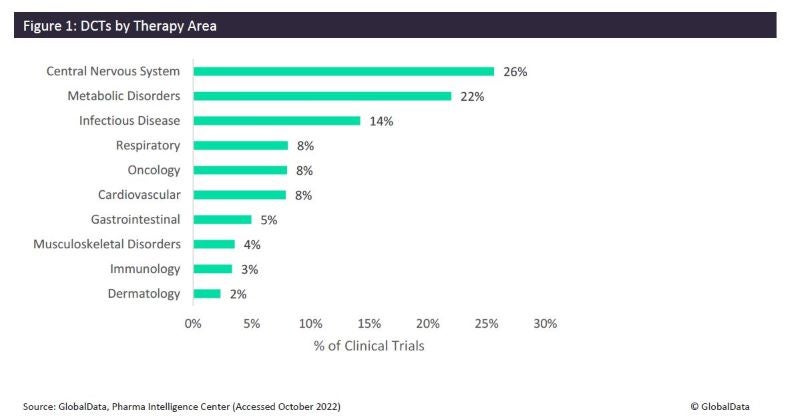
The central nervous system (CNS) is the most researched therapy area for DCT trials, with 26% of trials, followed by metabolic disorders at 22%. DCT usage has become increasingly popular in the majority of indications. Within CNS trials, remote patient monitoring (use of sensors or trackers) has become increasingly common. Some indications are more easily remotely monitored than others. For example, there may be fewer home measurement trackers or sensors in oncology compared to other indications, which may justify the reduced number in DCT trials for oncology. Some indications have more challenges to overcome, such as the administration of therapeutics (such as intravenous medication) or the need for in-person monitoring.