From early March 2020, around 1,000 organisations supporting clinical trials as a sponsor, collaborator or contract research organisation publicly announced disruptions to planned and ongoing clinical trials in their press releases, Securities and Exchange Commission filings and clinical trial registries, as well as on social media. Companies had to delay the initiation of planned trials or withdraw them completely, suspend enrolment in ongoing trials or terminate those trials altogether. GlobalData has dynamically tracked these disrupted trials and organisations, along with trials that have resumed activity since disruption.
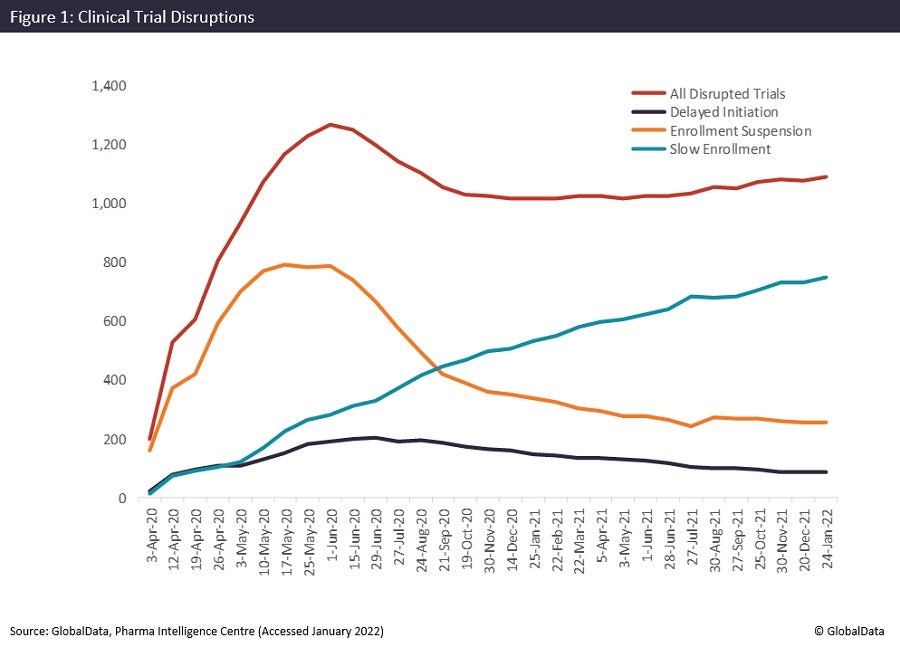
In June 2020, the number of total disrupted trials started falling slowly, as shown in Figure 1, but this levelled off in October 2020. The majority of current trial disruptions are in trials impacted by slow enrolment, which continue to increase this year. Trials impacted by enrolment suspension and delayed initiation maintain a slight downward trajectory. This suggests trials that were planned before being delayed, or that had initiated enrolment with chosen sites and investigators but were then suspended due to Covid-19, are having more success resuming.
Many hospitals that serve as trial sites were inundated with Covid-19 patients and may still not be available due to the emergence of the new Covid-19 variant, Omicron, which is more contagious and has caused spikes in confirmed cases in some countries. Likewise, many investigators may have been reassigned to Covid-19 drug discovery trials or treating Covid-19 patients, while the activation of sites for non-Covid-19 trials has, in some cases, been deprioritised. As the number of trials that have been impacted by slow enrolment continues to increase, this continues to be an issue. There is also a high risk to subjects in a clinical trial who have a serious chronic or acute condition that affects their immune system, giving them a greater chance of contracting Covid-19 and making them unwilling to enrol in a clinical trial.