Acasti Pharma, a late-stage biopharma company with drug candidates addressing rare and orphan diseases, has announced that WuXi Clinical Development will conduct its STRIVE-ON Phase III safety trial for GTX-104, a novel injectable nimodipine formulation for intravenous infusion (IV) that addresses high, unmet medical needs for aneurysmal subarachnoid haemorrhage (aSAH).
China-based WuXi Clinical is a global contract research organisation and a wholly owned subsidiary of WuXi AppTec.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
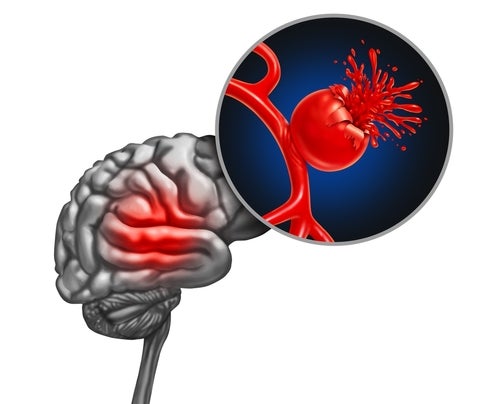
A rare disease, aSAH causes bleeding in the space that surrounds the brain. It is usually caused by leaking and bursting from weak areas in blood vessels on the brain’s surface.
STRIVE-ON (Safety, Tolerability, Randomised, IV and Oral Nimodipine) will assess the safety and tolerability of IV nimodipine compared to oral nimodipine in hospitalised patients.
Acasti submitted its Phase III safety study protocol for GTX-104 to the US Food and Drug Association (FDA) in May 2023 while making preparations with WuXi for kickoff.
There will be approximately 100 patients enrolled in the trial at around 25 hospitals in the US. The primary endpoint will be safety, measured as adverse effects including hypotension between the two groups.
GTX-104 will be administered as a continuous IV infusion of 0.15 mg/hour, and a 30-minute IV bolus of 4 mg every 4 hours. Oral nimodipine will be administered at 60 mg every 4 hours.
Both groups will receive GTX-104 or oral nimodipine for up to 21 consecutive days and safety and efficacy will be evaluated from the beginning of treatment through to a 90-day follow-up period.
“WuXi Clinical is a leader in the CRO industry with a strong track record in successfully helping pharmaceutical companies develop innovative therapies,” said Prashant Kohli, CEO of Canada-based Acasti. “Importantly, they are one of the few CROs with significant experience in aSAH and rare diseases, making them the ideal partner for conducting the STRIVE-ON Phase 3 safety trial for GTX-104.”





