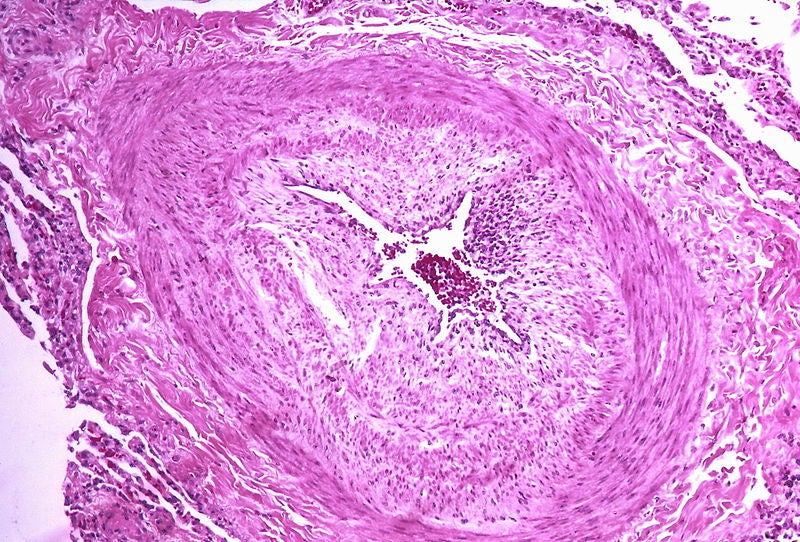
Acceleron Pharma has reported that the Phase II PULSAR study of sotatercept achieved its primary and major secondary endpoints for the treatment of pulmonary arterial hypertension (PAH).
Sotatercept is an investigational, selective ligand trap for the TGF-beta superfamily. It is intended to rebalance bone morphogenetic protein receptor type II (BMPR-II) signalling, a key molecular driver of PAH.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The Phase II study was conducted to assess the safety and efficacy of a 0.3mg/kg and 0.7mg/kg subcutaneous dose of the drug given every 21 days in combination with stable background therapies specific to PAH.
Enrolment for the trial was completed in June last year. The trial’s primary endpoint was the change in pulmonary vascular resistance (PVR) from baseline over 24 weeks.
Secondary endpoints included six-minute walk distance (6MWD), the change in amino-terminal brain natriuretic propeptide (NT-proBNP), and WHO functional class.
Sotatercept led to a statistically significant decrease in PVR compared to placebo over a 24-week treatment period.
A statistically significant improvement was observed in 6MWD, NT-proBNP and the WHO functional class when treated with sotatercept. The drug was generally well tolerated in the trial, with adverse events consistent with prior data in other diseases.
Acceleron Pharma president and CEO Habib Dable said: “PAH is a debilitating disease of high unmet medical need, so we’re encouraged by these data that signal that sotatercept could deliver added benefit to patients.
“We look forward to upcoming interactions with health authorities as we plan to globally develop and, if approved, commercialise sotatercept in PAH.”
Trial participants were eligible for an 18-month extension period after the six-month treatment period. Currently, 97 out of the 106 participants are participating in the extension period.





