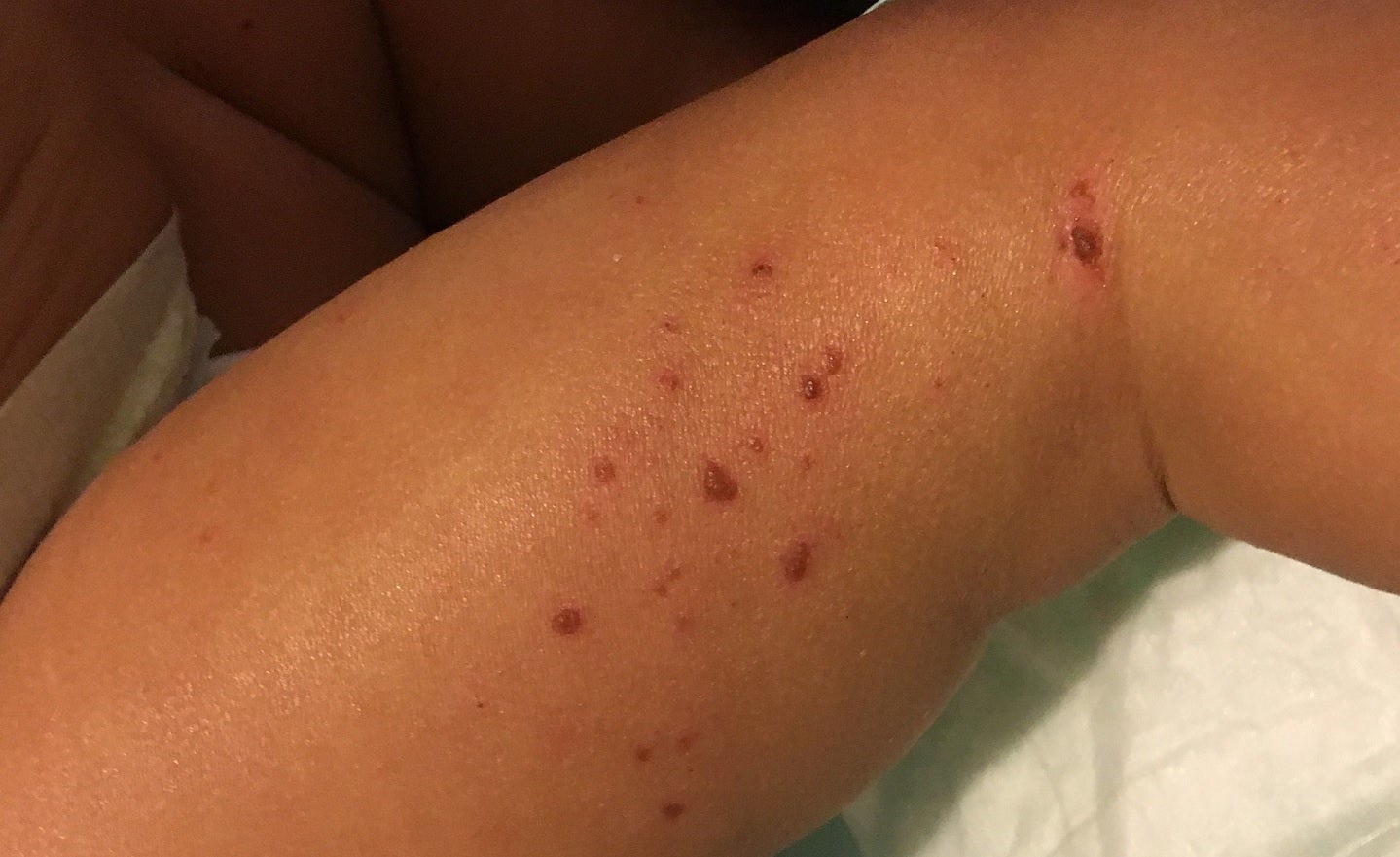
Clinical-stage biotechnology company Biosion USA has opened an investigational new drug (IND) after the US Food and Drug Administration review to commence a Phase II clinical trial of BSI-045B to treat atopic dermatitis (AD).
The trial has been designed for evaluating BSI-045B’s efficacy as monotherapy and along with Dupixent for further increasing the therapeutic effectiveness for AD patients.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The high-affinity, humanised monoclonal antibody (mAb) BSI-045B targets thymic stromal lymphopoietin (TSLP), which is a cytokine implicated in the pathogenesis of asthma, AD, as well as other eosinophilic and Th2 immune-related diseases.
In addition to excellent pharmacokinetic (PK), favourable safety profile, the monoclonal antibody showed potential to be a first-in-class treatment for AD, because of its single dose activity in an AD patients’ cohort, in a Phase I trial.
Biosion USA president and Biosion chief operating officer Dr Hugh Davis said: “BSI-045B is a next generation anti-TSLP mAb with high affinity and bioactivity that has the potential to offer greater therapeutic potential for patients with AD.
“Initiating the Phase II clinical trial is an important step toward our goal of developing antibody-based therapeutics for patients with unmet medical needs worldwide.”
Currently, Chinese pharmaceutical firm CTTQ, the collaboration partner of Biosion, is also conducting a Phase II clinical trial of BSI-045B to treat severe uncontrolled asthma in China.
CTTQ holds the rights to develop and commercialise BSI-045B (TQC2731) in the country.





