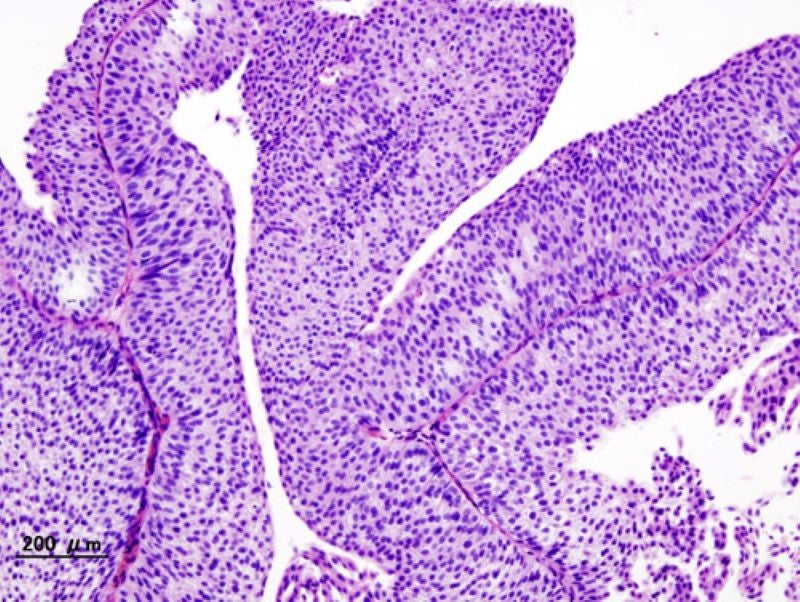
UroGen Pharma has reported the latest findings from the Phase IIb OPTIMA II clinical trial of its investigational drug, UGN-102 (mitomycin), to treat low-grade, intermediate-risk, non-muscle invasive bladder cancer (LG-IR-NMIBC).
The single-arm, multicentre, open-label trial analysed the safety and efficacy of the intravesical solution of UGN-102 in LG-IR-NMIBC patients.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
According to the trial findings, UGN-102 offered a median duration of response (DOR) of 24.4 months in trial subjects.
DOR was evaluated as the time from the reported complete response (CR) in the trial to the recurrence of disease or mortality, or the last adequate disease analysis (for participants who remained in CR).
Seven subjects remained in complete response and six had LG disease recurrence.
One trial subject progressed to high-grade disease while another patient withdrew consent but remained in CR.
At three months, a CR of 65% was reported.
At 12 months from treatment commencement or nine months after CR, Kaplan-Meier analysis estimated DOR was found to be 72.5%, without attaining median DOR.
UGN-102 was also reported to be well tolerated, with adverse events seen to be mild to moderate in nature.
Created to remove tumours non-surgically, UGN-102 is presently in the Phase III development stage.
UroGen chief medical officer Mark Schoenberg said: “UGN-102 uses a similar combination with a simpler delivery method to our currently approved chemoablative medicine and has showed a similar durability of response in LG-IR-NMIBC.
“If approved, UGN-102 would be the only primary non-surgical treatment option for patients with LG-IR-NMIBC who often recur within one year of receiving surgery and continue to need repetitive surgeries for the rest of their life.”
In February this year, the company began the Phase III ENVISION trial of UGN-102 for an intravesical solution for treating LG-IR-NMIBC.





