ADYNOVI® is an intravenous infusion indicated for the treatment of adults and adolescents with haemophilia A.
The drug was developed by US-based biopharmaceutical firm Baxalta, which was acquired by Shire in June 2016.
ADYNOVI® was formerly approved as ADYNOVATE® by the US Food and Drug Administration (FDA) in November 2015. Shire also submitted a marketing authorisation application (MAA) to the European Medicines Agency (EMA) in March 2016.
The drug received marketing authorisation approval from the European Commission (EC) in January 2018. The approval covers all 28 countries in the EU, as well as Iceland, Liechtenstein and Norway.
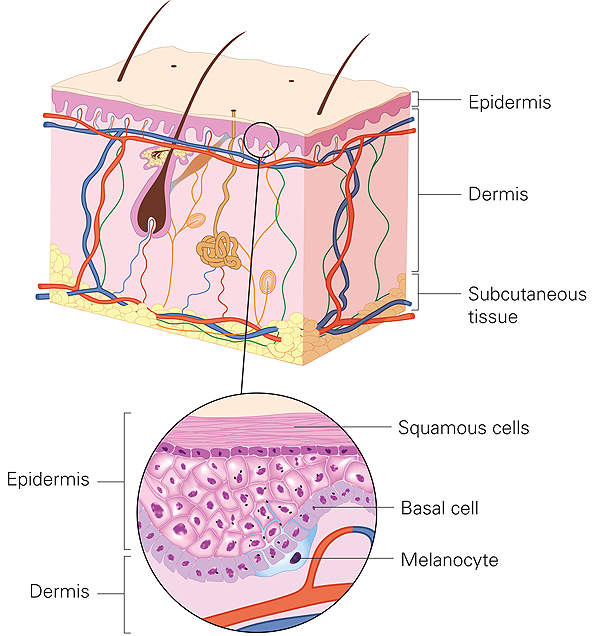
Haemophilia A causes and symptoms
Haemophilia A is a blood clotting disorder caused by the lack of anti-hemophilic factor (AHF), which is a protein in blood that aids clotting.
The disease is genetic and inherited as an X-linked recessive trait. However, there are cases that occur due to spontaneous mutations. Haemophilia A mostly affects the male population as they have a single X-chromosome.
The main symptoms of the disease are spontaneous or extended periods of bleeding following injuries or surgery. The disease affects joints or muscles, and can cause pain, chronic swelling, deformity, reduced mobility and long-term joint damage.
The disease is prevalent in approximately one in every 5,000 male births. More than 20,000 people in the US are living with the condition, according to the US Centres for Disease Control and Prevention (CDC).
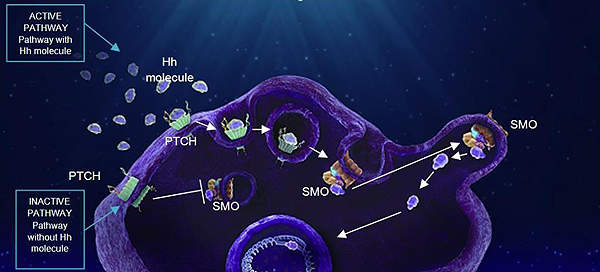
ADYNOVI’s mechanism of action
ADYNOVI® is a PEGylated form of recombinant anti-haemophilic factor (ADVATE) and contains rurioctocog alfa pegol.
The drug replaces the missing AHF protein required for effective haemostasis and helps blood to clot.
The twice-weekly regimen drug is available as a powder and solvent, which are mixed together to make an injectable solution.
Clinical trials on ADYNOVI
The EC’s approval for ADYNOVI® was based on data from three Phase III clinical trials on haemophilia A patients.
The first trial was a prospective, global, multi-centre, open-label, non-randomised study that enrolled 137 patients aged from 12 to 65 years. Out of the 137 patients, 120 were administered with prophylactic treatment of ADYNOVI® as a twice-weekly regimen, while 17 were administered with ADYNOVI® as an on-demand treatment.
The median annualised bleed rate (ABR) in the prophylaxis arm was 1.9, compared to 41.5 in the on-demand treatment arm.
The second prospective, uncontrolled, open-label, multi-centre study enrolled 66 patients aged 12 years and younger. Participants were administered with ADYNOVI® as a twice-weekly regimen for six months. More than 37% of patients had no bleeding episodes and 72% experienced no joint bleeding episodes during the treatment period.
A study was also conducted on the peri-operative management of haemostasis. The study recruited 15 subjects that had undergone 11 major surgeries and four additional minor surgeries. The perioperative haemostatic efficacy of ADYNOVI® was rated as excellent in all the major and minor surgeries.
The most common adverse reactions reported in patients during the clinical studies were headache nausea, diarrhoea and rashes.