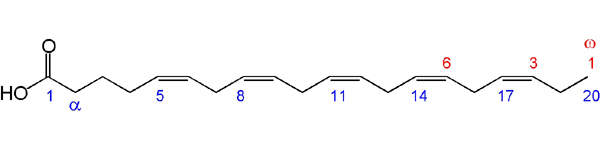
Under development by Amarin Corporation, AMR101 is a partially synthetic omega-3 fatty acid-based drug. It contains the purest form of the active ingredient, ethyl ester of eicosapentaenoic acid (ethyl-EPA).
AMR101 is being developed for the treatment of hypertriglyceridaemia, which is a key cause for cardiovascular diseases. Phase III clinical trials were completed in April 2011; Amarin plans to submit a new drug application for AMR101 to the US Food and Drug Administration (FDA) in the third quarter of 2011.
Hypertriglyceridaemia – cause and effect
Hypertriglyceridaemia is described as a condition in which triglycerides (a type of fat found in the blood) are present in high levels in the body. Triglycerides are produced from food and are also manufactured by the liver.
Hypertriglyceridaemia is known to increase the risk of cardiovascular diseases and cause a range of lipid disorders such as dyslipidemia.
Hypertriglyceridaemia can intensify with age and is more prevalent in people who are overweight or obese. It can also be caused by uncontrolled diabetes mellitus and a sedentary lifestyle. If the cause of the disease is genetic then it is known as familial hypertriglyceridaemia.
In the US, it is estimated that about 27 million individuals have high triglyceride levels and are at risk of developing cardiovascular diseases. They are also at the risk of developing other metabolic disorders such as diabetes and obesity.
AMR101 – ultra pure ethyl-EPA
In clinical studies the active ingredient in AMR101, ethyl-EPA, has shown its effectiveness in lowering high triglyceride levels. Irrespective of the degree of severity, AMR101 is effective in reducing plasma triglycerides in hypertriglyceridaemia patients.
Studies have also shown the safety and efficacy of AMR101 on a number of other elements in the body such as cell membrane composition and regulation of glucose metabolism. It has also been shown to be effective as an anti-inflammatory mechanism.
AMR101 has also been tested for central nervous system disorders such as Huntington’s disease. A Marketing Authorization Application for the treatment of the disease is pending with the European Medicines Agency following positive results from Phase III clinical trials. AMR101 has also received Orphan Medicinal Product designation in the EU for the treatment of the disease.
Global clinical trials for AMR101
In January 2008, Amarin was granted regulatory approval to initiate Phase IIa trials with AMR101 for age-associated memory impairment. The randomised, double-blind, placebo-controlled trial was carried out in the UK and enrolled 96 patients. It was initiated following positive results from pre-clinical studies in memory and cognition. The drug met all the primary objectives of the trial.
In July 2008, Amarin began Phase III development of AMR101 for the treatment of hypertriglyceridaemia. The company launched two Phase III clinical studies, the MARINE and ANCHOR studies. In January 2010, the first patients for the two studies were enrolled by the company. The studies were conducted under a Special Protocol Agreement with the FDA. The agreement covered the design, endpoints and statistical analysis of the Phase III clinical studies.
The MARINE study was a placebo-controlled, randomised, double-blind trial that recruited 229 patients. The 12-week study was conducted in North and Central America, Europe, India and South Africa, and was designed to assess the safety and efficacy of AMR101 in patients with high triglyceride levels.
In November 2010, results of the MARINE study were announced, and the drug was confirmed to have met both the trial’s primary and secondary endpoints. The study also showed that there was statically no increase in low-density lipoprotein cholesterol (LDL-C) levels. The safety profile of AMR101 was also found to be similar to placebo.
The ANCHOR study was a placebo-controlled, randomised, double-blind trial that involved 702 patients. The 12-week study assessed the safety and efficacy of AMR101 in individuals with high triglyceride levels who were undergoing statin therapy. It was conducted across various centres in the US.
Results of the ANCHOR study, announced in April 2011, indicated that AMR101 is effective in statistically decreasing LDL-C levels. The drug met all of the trial’s primary and secondary endpoints.
Marketing commentary
It is estimated that the number of hypertriglyceridaemia and dyslipidaemia cases in the US is more than 100 million. The market for these treatments is estimated to be more than $25m and predominantly includes treatments by statin therapies.
The non-statin market is growing steadily and represents a new approach towards treating such diseases. Instead of focusing on reducing low-density lipoprotein cholesterol, a key factor in causing dyslipidaemia, treatments are moving towards other factors such as regulation of high-density lipoprotein cholesterol and triglycerides.
If approved AMR101 will be the first omega-3 based drug in the market for treating both high triglycerides levels and mixed dyslipidaemia. At the moment Lovaza is the only omega-3 based drug approved in the market for the treatment of high triglycerides levels.
AMR101 will also be entitled to ten years of market exclusivity in the EU following its designation as an Orphan Medicinal Product. This considerably improves the drug’s prospects in the market for the treatment of Huntington’s disease.