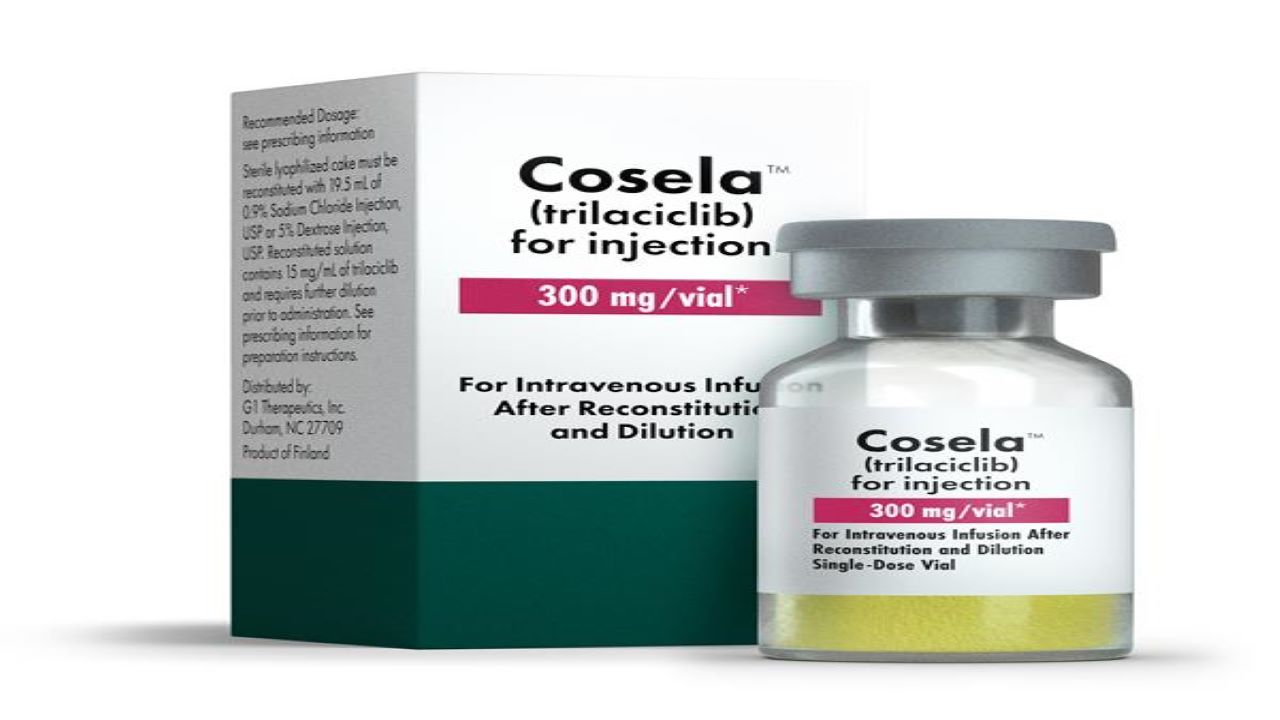
COSELA™ (trilaciclib) is the first approved myeloprotection therapy indicated to reduce the occurrence of chemotherapy-induced bone marrow suppression in adult patients.
Developed by G1 Therapeutics (GTHX), a US-based clinical-stage biopharmaceutical company, the drug is available in a single-dose vial as a sterile, preservative-free, yellow lyophilised cake in a 300mg dosage strength for intravenous administration.
In June 2020, G1 Therapeutics signed a three-year co-promotion agreement with German pharmaceutical firm Boehringer Ingelheim (BI) to jointly promote trilaciclib for the treatment of small cell lung cancer in the US and Puerto Rico.
Under the agreement, G1 Therapeutics will lead marketing, market access and medical engagement initiatives for COSELA while Boehringer Ingelheim will undertake salesforce engagements.
In August 2020, China-based Simcere Pharmaceutical Group was granted the development and commercialisation rights of the drug in all indications for Greater China.
COSELA approvals
The New Drug Application (NDA) for trilaciclib was submitted to the US Food and Drug Administration (FDA) in June 2020 and granted priority review in August 2020.
In February 2021, the FDA approved trilaciclib to reduce chemotherapy-induced myelosuppression in extensive-stage small cell lung cancer (ES-SCLC) patients, prior to chemotherapy treatments involving platinum-etoposide or topotecan options. The FDA also bestowed breakthrough therapy designation to trilaciclib in August 2019.
Chemotherapy-induced myelosuppression causes and symptoms
Myelosuppression, also known as bone marrow suppression, is a chemotherapy-induced bone marrow damage condition that lowers blood cell production.
Although chemotherapy drugs are used to destroy cancer cells, they can also cause damage to healthy cells in the bone marrow such as hematopoietic stem and progenitor cells (HSPCs), which produce white blood cells, red blood cells and platelets.
Myelosuppression is a side effect of chemotherapy and occurs when the hematopoietic stem and progenitor cells are damaged by chemotherapy treatment, thereby suppressing the ability of bone marrow to produce blood cells.
The common symptoms associated with myelosuppression include fatigue, shortness of breath, and dizziness. Myelosuppression can also lead to serious blood cell diseases such as anaemia, neutropenia and thrombocytopenia.
Trilaciclib mechanism of action
Trilaciclib is a transient and competitive inhibitor of cyclin-dependent kinases 4 and 6 (CDK4/6). The drug delivers a myeloprotective therapy against chemotherapy-induced bone marrow suppression by inhibiting CDK4/6 that regulates cell cycle.
By inhibiting CDK4/6, trilaciclib temporarily and reversibly induces G1 cell cycle arrest in hematopoietic stem and progenitor cells (HSPCs) and prevents transition to the synthesis phase (S phase) of cell cycle, thus protecting the HSPCs from the damaging effects of chemotherapy and maintaining the normal function of the bone marrow.
Clinical trials on COSELA
COSELA’s FDA approval was based on the outcome of three randomised, double-blind, placebo-controlled clinical trials in patients with extensive-stage small cell lung cancer. The effectiveness of drug was evaluated in combination with carboplatin-etoposide, with or without atezolizumab and topotecan chemotherapy.
The studies randomly enrolled 245 patients to receive either intravenous (IV) trilaciclib or placebo prior to the start of chemotherapy.
The primary endpoints of the studies were the percentage of patients with severe neutropenia and its duration during the first chemotherapy cycle.
The trials demonstrated clinical reduction in the duration and severity of neutropenia among ES-SCLC patients who received trilaciclib before chemotherapy.
A positive impact on red blood cell transfusions and other myeloprotective measures was also observed.
The most frequent side effects observed in the patients during the clinical trials were fatigue, hypophosphatemia, hypocalcaemia, hypokalaemia, headache, high aspartate aminotransferase levels and pneumonia. More than 3% of the patients who received COSELA experienced serious adverse reactions, including respiratory failure, haemorrhage and thrombosis.