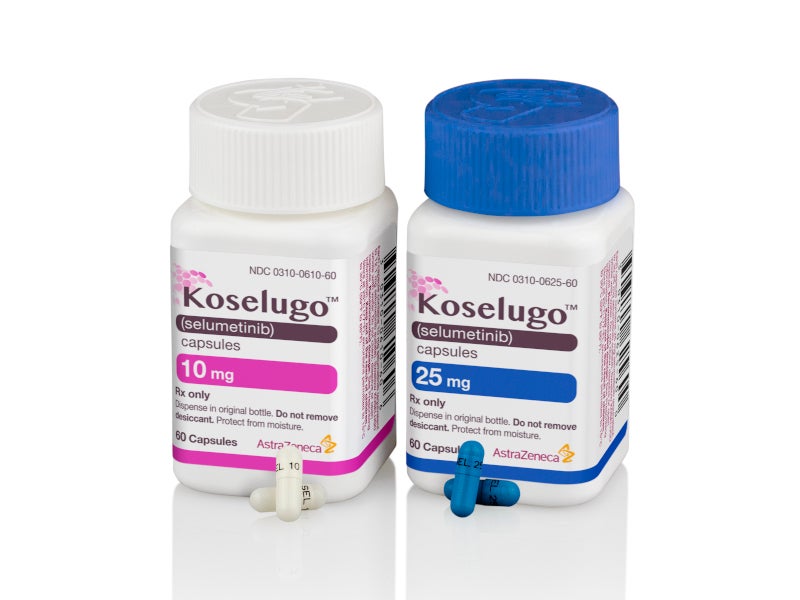
Koselugo® (selumetinib) is an oral mitogen-activated protein kinase (MEK) inhibitor indicated for the treatment of neurofibromatosis type 1 (NF1), a rare and incurable genetic condition.
The drug was developed and commercialised by AstraZeneca and Merck (MSD) under a licensing agreement.
AstraZeneca’s subsidiary Alexion, AstraZeneca Rare Disease, is currently overseeing the development and commercialisation of Koselugo.
Koselugo (selumetinib) is available as oral capsules with a recommended dose of 25mg/m2 administered twice daily.
Regulatory approvals
Koselugo received EU orphan designation in August 2018 and orphan drug status from Swissmedic in December 2018. It was granted breakthrough therapy designation by the US Food and Drug Administration (FDA) in April 2019 and rare paediatric disease designation in December 2019.
The FDA accepted a new drug application for selumetinib and granted priority review for Koselugo in November 2019.
Koselugo was approved by the FDA in April 2020 for treating NF1 with symptomatic and irremediable plexiform neurofibromas (PN) in paediatric patients older than two years.
Subsequently, the FDA approved Koselugo for paediatric patients aged one year and older based on SPRINT Phase II Stratum I and SPRINKLE studies in September 2020.
MSD submitted a marketing authorisation application for Koselugo to the European Medicines Agency in early 2020.
Koselugo received conditional approval in the EU in June 2021 to treat PN NF1 in paediatric patients aged three years and above.
The drug also received temporary authorisation in Switzerland in July 2022.
The Japanese Ministry of Health, Labour and Welfare approved Koselugo® as the first medicine to treat paediatric patients aged three years and older with PN NF1 in September 2022.
In May 2023, the National Medical Products Administration in China approved the drug for the same indication.
The drug was approved by Health Canada as the first and only therapy for the treatment of NF1 PN in paediatric patients aged two years and above in April 2023. It was also approved in Australia for the same indication in August 2024.
Koselugo was approved by the European Commission (EC) and the FDA for the treatment of symptomatic, inoperable PN in adult patients with NF1 in October and November 2025, respectively, based on results from the KOMET Phase III trial.
NF1 causes and symptoms
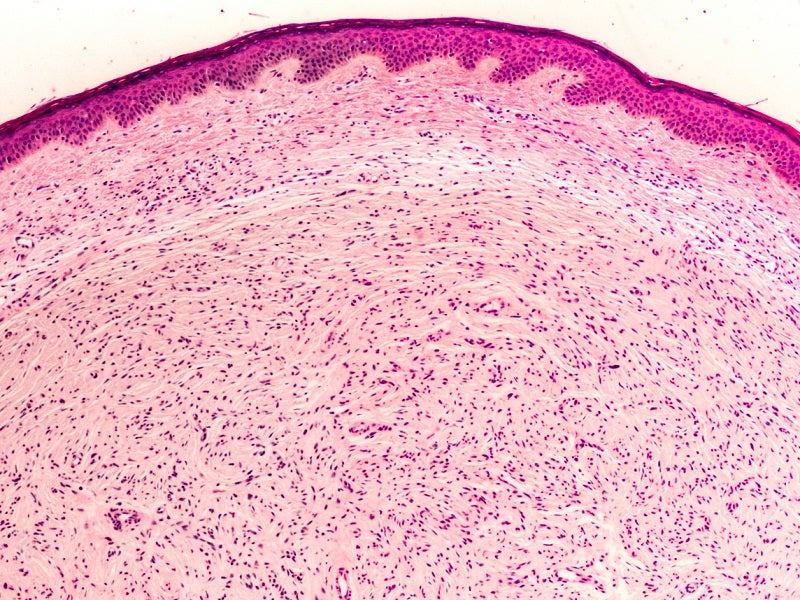
NF1 is a rare and untreatable genetic disease that causes irregular skin colour (pigmentation) and development of benign tumours on nerves, skin (neurofibromas), brain and other body parts.
NF1 is also known as Von Recklinghausen’s disease, peripheral plexiform neurofibromas (NF) and Von Recklinghausen neurofibromatosis.
It is caused by mutations in the NF1 gene found on chromosome 17. The NF1 gene regulates the production of neurofibromin protein, which inhibits the uncontrolled tumour growth.
Common signs and symptoms include short stature, abnormal large head size (macrocephaly), skeletal malformations including a lateral curvature of the spine (scoliosis), speech difficulties, hyperactivity, high blood pressure and coloured bumps on the iris (Lisch nodules).
NF1 is mainly observed during the early years of childhood and is anticipated to reduce life expectancy by up to 15 years.
Selumetinib mechanism of action
Selumetinib is a mitogen-activated protein kinases 1 and 2 (MEK1/2) inhibitor. The MEK1/2 proteins regulate the upstream extracellular signal-regulated kinase (ERK) pathway.
Both MEK and ERK are critical components of the RAS-regulated RAF-MEK-ERK pathway, often activated in various types of cancers.
In NF1, MEK activity is abnormally increased, leading to uncontrolled tumour cell growth and the development of PN. By inhibiting MEK1/2, Koselugo helps to curb tumour cell proliferation and, consequently, slows the progression of PN.
Clinical trials on Koselugo
The US FDA, the EU, Canada, Japan and China approvals of Koselugo to treat paediatric patients aged two years and above came from the non-randomised, multicentre, open-label, single-arm Phase II clinical trial, SPRINT Stratum 1.
The SPRINT Stratum 1 trial was coordinated by the National Cancer Institute’s (NCI) Centre for Cancer Research, Paediatric Oncology Branch, and sponsored by the NCI Cancer Therapy Evaluation Programme.
In the SPRINT clinical trial, 99 eligible patients received selumetinib 25mg/m2 orally every 12 hours daily during each 28-day cycle until unacceptable toxicity, patient withdrawal or disease progression.
The primary endpoint measure in the SPRINT Stratum 1 trial was overall response rate (ORR), defined as the percentage of patients with confirmed complete response (vanishing of the target PN) or partial response of at least 20% reduction in PN volume on MRI that was confirmed on a subsequent MRI within 3–6 months.
ORR was 66%, and all patients (n=33) witnessed a partial response. No patients had complete tumour disappearance while 82% of patients (n=27) had a response enduring for more than 12 months.
Among the serious adverse reactions observed during Phase II SPRINT Stratum 1 trial were ocular toxicity, skin toxicity, increased creatinine phosphokinase, risk of bleeding and foetal harm.
The FDA approval of Koselugo for paediatric patients aged one year and older was based on the SPRINKLE Phase I/II, single-arm, open-label study, aimed at demonstrating the efficacy of selumetinib oral granule formulation in children aged between one and seven years.
The study enrolled 36 patients, divided into two cohorts, 17 patients aged one to three years and 15 patients aged four to seven years, to analyse drug exposure profiles.
Assessment of the results revealed that selumetinib reached peak concentration in a similar time frame across both age groups, indicating comparable pharmacokinetics. This observation supports the analytical rationale for extrapolating efficacy and safety data from the older cohort to the younger cohort.
Further clinical trials on Koselugo
The FDA and EC’s approvals of Koselugo to treat symptomatic, inoperable PN in adult patients with NF1 were supported by findings from the Phase III KOMET trial.
The randomised, double-blind, placebo-controlled, multicentre study examined the efficacy and safety of Koselugo in 145 adults from 13 countries.
The primary endpoint of the trial was a confirmed ORR by cycle 16, as determined by independent central review. Secondary endpoints included assessments of PN-related pain reduction and improvements in health-related quality of life at cycle 12.
Participants were randomised in equal numbers to receive either selumetinib (n=71) or placebo (n=74) over 12 cycles, each lasting 28 days.
The study’s results revealed that Koselugo achieved a statistically significant ORR of 20% by cycle 16 versus 5% with placebo, with the difference reaching statistical significance (p=0.01).
Moreover, 86% of patients treated with selumetinib achieved an observed duration of response of at least six months.
After the initial 12 cycles, individuals receiving a placebo were switched to Koselugo, while those initially treated with Koselugo continued treatment for an additional 12 cycles.