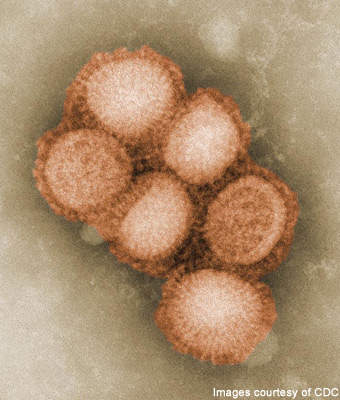
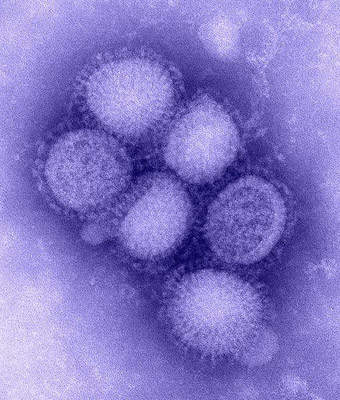
Developed by Sinovec Biotech to provide protection against the H1N1 virus, also known as swine flu, Panflu.1 is a single-shot vaccine. The vaccine, developed in response to the worldwide outbreak of the H1N1 virus in April 2009, was released for delivery in September 2009.
Sinovec received the first order of 3.3 million doses from the government of China on 4 September 2009. Subsequently, on 30 September 2009, the Chinese Ministry of Industry and Information Technology placed an additional order of three million doses.
By September 2009, over 100,000 Beijing citizens had been administered with Panflu.1. The vaccine has been approved to be distributed in Mexico until October 2014. It is the first China-made vaccine to get such approval in Mexico.
Available as two doses in one vial, Panflu.1 is suitable for people aged three to 60. A revised production licence issued by the Chinese State Food and Drug Administration in November 2009 expanded the target audience to include 60 year olds. It also extended the vaccine’s shelf life from six months to a year.
H1N1 pandemic influenza
By mid-2009, influenza H1N1 2009 had been declared a global pandemic. Starting in Veracruz, Mexico, the virus spread rapidly with over 75,000 laboratory confirmed cases and over 4,500 reported deaths globally as of October 2009.
On 11 June 2009, the World Health Organization increased its pandemic alert to six, the highest level, indicating that it was a global pandemic.
While the majority of the affected have shown only mild symptoms such as fever, sore throat, cough, headache, muscle or joint pains, nausea, vomiting or diarrhoea, a handful of patients have experienced serious symptoms. Those with asthma, diabetics, the obese, people with heart diseases, the immunocompromised, children with neurodevelopmental conditions and pregnant women have been the worst affected.
The H1N1 virus develops resistance to the antiviral oseltamivir (Tamiflu). To date, the 31 pandemic H1N1 influenza viruses that have been detected worldwide demonstrate the same H275Y mutation that shows resistance to oseltamivir, but not to the antiviral zanamivir (Relenza). Globally, over 10,000 clinical specimens of the virus have been tested and declared to be oseltamivir sensitive.
Preventing infection
Panflu.1 contains a killed or weakened part of the germ that is responsible for infection. Since the germ has been killed or weakened prior to being used for making the vaccine, it cannot infect a person.
When a person receives a shot of Panflu.1, the body reacts by creating antibodies. Acting as body defenders, the antibodies destroy the germs.
Clinical trials demonstrate Panflu.1’s efficacy
The safety and efficacy of the vaccine was tested in a clinical trial undertaken by the Beijing Centre for Disease Control from 22 to 25 July 2009. Organised by China’s Center for Disease Control, the clinical trial was conducted on 1,614 volunteers, including 101 elderly, 706 adults, 404 youths and 403 children.
During the trial, three different types of vaccine – split viron vaccine, split viron vaccine with adjuvant and whole viron vaccine with adjuvant – were tested. The volunteers were dosed with 5–30ug of Panflu.1 within an immunisation schedule of zero to 21 days. The first shot of the vaccine was given to the volunteers by 25 July 2009.
The volunteers showed no signs of severe adverse reactions during the period of observation. The adverse event rate with Panflu.1 was recorded at 11.8%, similar to vaccines meant for seasonal influenza. Only mild and temporary adverse events were demonstrated, with the most severe event being the pain felt while the shot of the vaccine was given.
Results from the clinical trial indicated that the vaccine provided protection from the H1N1 virus after only one shot. To develop the expected levels of immunity, the other existing swine flu vaccines are required to be administered in two doses.