Sovaldi (sofosbuvir), developed by Gilead Sciences (Gilead), is an oral nucleotide analogue inhibitor indicated for the treatment of chronic hepatitis C (CHC) infection.
Gilead submitted a new drug application for Sovaldi to US Food and Drug Administration (FDA) in April 2013. The FDA approved Sovaldi for treatment of patients with hepatitis C virus (HCV) genotypes 1, 2, 3 or 4 infection in December 2013. Sovaldi is also approved in Australia, Canada, New Zealand and Switzerland.
Gilead also received positive opinion for the marketing authorisation application (MAA) for Sovaldi for the treatment of CHC infectionfrom the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) in November 2013.
The once-daily fixed-dose combination of the NS5A inhibitor ledipasvir (LDV) 90mg and the nucleotide analogue polymerase inhibitor sofosbuvir (SOF) 400mg drug is currently under an accelerated review procedure of the EMA.
In January 2014, Sovaldi SOF 400mg obtained marketing authorisation from European Commission (EC) as a single agent once-daily oral drug for the treatment of CHC genotypes 1-6 infection, in combination with other antiviral agents (ribavirin (RBV) and pegylated interferon alpha (peg-IFN).
Applications for marketing approval of Sovaldi are also under review in Japan and Turkey.
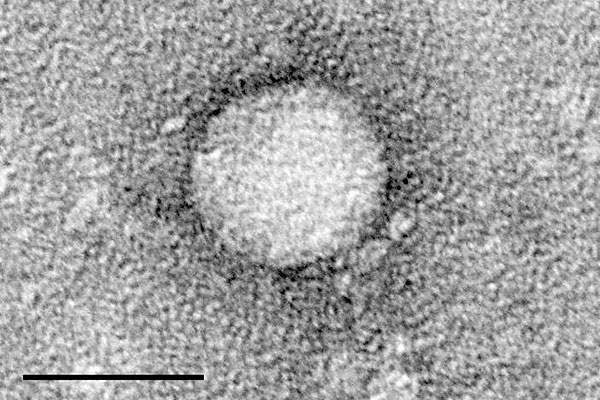
Hepatitis C causes
Hepatitis C is an infectious disease affecting the liver. It is caused by the hepatitis C virus (HCV), which can lead to scarring of the liver and ultimately to cirrhosis. The infectious disease is estimated to affect four million people in the US every year.
Sovaldi’s mechanism of action
Sovaldi contains oral nucleotide analogue inhibitor of the hepatitis C virus (HCV) polymerase. The drug is a direct-acting antiviral agent and interferes with the HCV lifecycle, restraining viral replication.
It can be incorporated into HCV RNA by the NS5B polymerase and acts as a chain terminator. The drug is available for oral use in the form of 400mg tablets.
Clinical trials on Sovaldi (sofosbuvir)
Gilead Sciences conducted Phase II and III clinical studies on Sovaldi on more than 3,000 patients. Sovaldi combination therapy was well-tolerated in all the clinical studies. Fatigue, headache, nausea, insomnia and anaemia were the most common adverse events found in 20% of patients receiving Sovaldi in combination with Peg-IFN / RBV during clinical studies.
The FDA approval for Sovaldi was based on data assimilated from four Phase 3 clinical trials known as NEUTRINO, FISSION, POSITRON and FUSION. Studies evaluated Sovaldi in combination with either RBV or RBV plus peg-IFN. Genotype 2 or 3 patients were enrolled in the FISSION, FUSION, and POSITRON clinical studies. Patients were administered with Sovaldi plus RBV. The NEUTRINO clinical study administered Sovaldi in combination with Peg-IFN / RBV in treatment naïve patients with genotypes 1, 4, 5 or 6.
Study results demonstrated that Sovaldi-based therapy was significantly more effective than historical controls or placebo, or non-inferior to currently available treatment options. Patients administered with Sovaldi achieved SVR12 rates of 50%-90%. No viral resistance to Sovaldi was detected among patients who relapsed following completion of therapy in all the Phase III studies.
The FDA also added data assimilated from two additional Phase III studies, VALENCE and PHOTON-1 to review Sovaldi.
Patients with genotype 3 HCV infection were enrolled in the VALENCE study. They were administered with Sovaldi and RBV for 24 weeks. Approximately 84% of patients in this trial achieved SVR12.
Patients with genotype 2 HCV infection were enrolled in the PHOTON-1 study. They were administered with Sovaldi and RBV for 12 weeks, and achieved SVR12 rates between 76% and 92% in the study.
Marketing commentary
Gilead Sciences is a leading biopharmaceutical company headquartered at Foster City in California, US. The company is engaged in the discovery, development and commercialisation of innovative therapeutics in areas of unmet medical need. Its operations are spread across North and South Americas, Europe, and Asia Pacific.
Other medications available for the treatment of chronic hepatitis C include Incivek developed by Vertex Pharmaceuticals, Mitsubishi Tanabe Pharma Corporation and Tibotec BVBA, and Victrelis (boceprevir) manufactured by Schering-Plough and Merck.