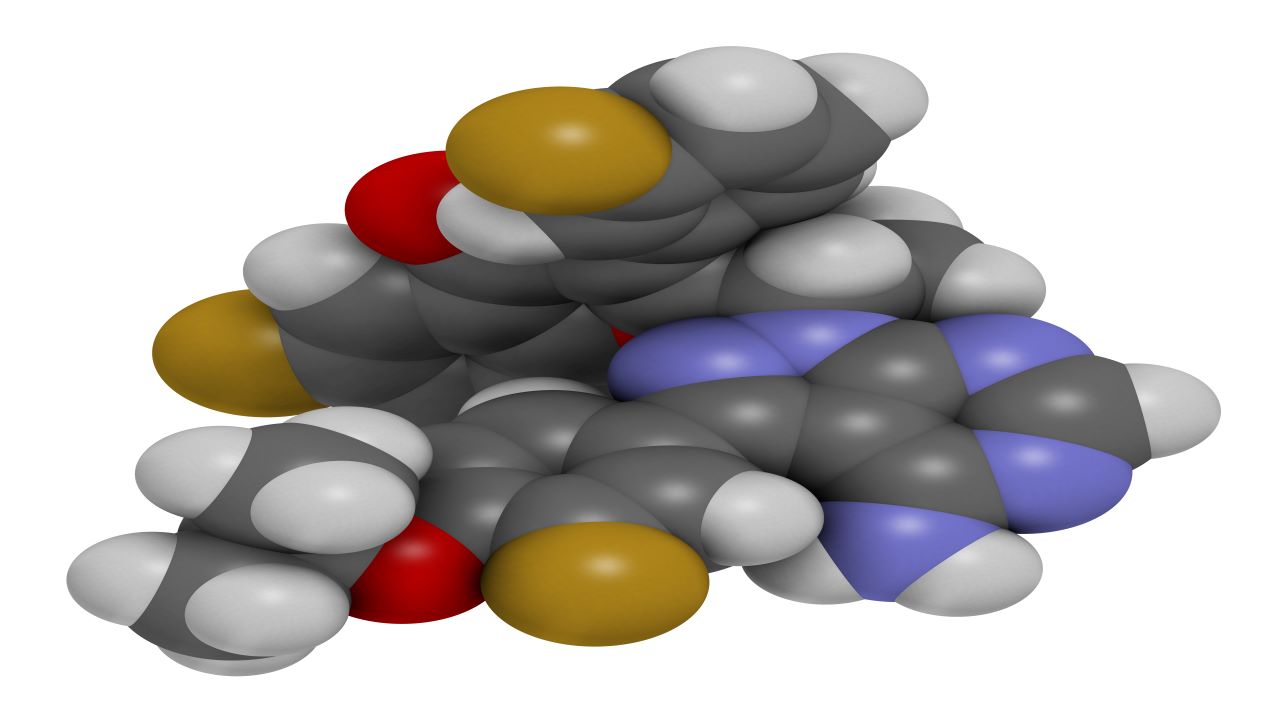
UKONIQ™ (umbralisib) is the first and only oral, dual inhibitor of phosphoinositide 3 kinase (PI3K) delta and casein kinase 1 (CK1) epsilon, indicated for the treatment of previously treated relapsed or refractory marginal zone lymphoma (MZL) and follicular lymphoma (FL) in adult patients.
Developed by TG Therapeutics, a clinical-stage biopharmaceutical company based in the US, UKONIQ is available as an oral once-daily, oval, green, film-coated tablets with a dosage strength of 200mg.
In August 2012, TG Therapeutics signed an exclusive global agreement with Swiss-based Rhizen Pharmaceuticals for the development and commercialisation of umbralisib, formerly known as TGR-1202.
TG Therapeutics obtained the global rights of umbralisib, excluding India, from Rhizen Pharmaceuticals, under a licensing agreement signed in September 2014. Rhizen Pharmaceuticals retains its commercialisation rights for India and also acts as the production and supply partner for the drug.
UKONIQ approvals
TG Therapeutics initiated rolling submission of a new drug application (NDA) for UKONIQ to the US Food and Drug Administration (FDA) in January 2020 and completed the same in June. The NDA was accepted by the FDA in August 2020.
The FDA granted accelerated approval to UKONIQ for the treatment of relapsed or refractory marginal zone lymphoma in adult patients previously treated with at least one anti-CD20 based therapy in February 2021.
The drug also secured accelerated approval from the FDA for the treatment of relapsed or refractory follicular lymphoma in adult patients who received at least three prior systemic therapies. UKONIQ also holds a breakthrough therapy designation (BTD) from the FDA for the treatment of MZL, as well as an orphan drug designation (ODD) for the treatment of MZL and FL.
Marginal zone lymphoma and Follicular lymphoma causes and symptoms
Marginal zone lymphoma, which accounts for nearly 10% of all non-Hodgkin lymphoma (NHL) cases, is the third most prevalent B-cell NHL (non-Hodgkin lymphoma). Follicular lymphoma is the second most prevalent form of NHL, which represents nearly 17% of all NHL cases. Approximately 8,200 new MZL and nearly 13,200 new FL cases were diagnosed in the US in 2020.
Both MZL and FL are a type of slow-growing NHLs that develop from B lymphocytes and are characterised by multiple recurrences and relapses.
Further, MZL is classified into three subtypes namely, extranodal MZL of the mucosal-associated lymphoid tissue (MALT), nodal marginal zone lymphoma (NMZL) and splenic marginal zone lymphoma (SMZL).
Follicular lymphoma is the second most prevalent form of NHL, which represents nearly 17% of all NHL cases. Approximately 8,200 new MZL and nearly 13,200 new FL cases were diagnosed in the US in 2020.
The aetiology of MZL is associated with autoimmune conditions and certain infections such as Helicobacter pylori and hepatitis C, which leads to an overstimulated immune system.
Umbralisib mechanism of action
Umbralisib is a selective inhibitor of PI3 kinase-delta, a delta isoform of phosphoinositide-3-kinases (PI3Ks). which is primarily expressed in the haematopoietic cells and malignant lymphoid disorders.
PI3 kinase-delta, a delta isoform of phosphoinositide-3-kinases (PI3Ks), plays a vital role in cell survival and proliferation, cell differentiation, intercellular trafficking and immunity.
Umbralisib also inhibits casein kinase 1 epsilon (CK1-epsilon), a regulator of oncoprotein translation. CK1-epsilon is associated with the growth and survival of lymphoma cells.
Clinical trials on UKONIQ
FDA approval of the drug was based on the outcome of an open-label, multi-centre, multi-cohort, phase two clinical trial, UNITY-NHL.
The study evaluated the safety and efficacy of UKONIQ in 117 patients with FL who had previously received at least two prior systemic therapies, including an anti-CD20 and an alkylating agent. It also enrolled 69 patients with MZL after at least one prior therapy, including an anti-CD20 containing regimen.
Patients were administered with an umbralisib 800mg oral tablet orally once daily until disease progression or unacceptable toxicity.
The primary efficacy outcome measure was an overall response rate (ORR), as reviewed by an independent review committee (IRC) using Revised International Working Group criteria.
MZL patients treated with umbralisib in the UNITY-NHL trial for MZL achieved an ORR of 49%, with 16% achieving complete responses (CR). FL patients achieved an ORR of 43%, with 3% achieving complete responses.
The most common adverse reactions observed during the trial were diarrhoea-colitis, nausea, increased creatinine, fatigue, neutropenia, musculoskeletal pain, anaemia, thrombocytopenia, upper respiratory tract infection, vomiting, abdominal pain, decreased appetite and rash.