Developed by Pfizer Global Research and Development, capravirine is a Non-Nuclesoide Reverse Transcriptase Inhibitor (NNRTI) that was under development for the treatment of patients infected with the Human Immunodeficiency Virus (HIV). It was indicated for patients with HIV/AIDS who had failed on currently available antiretroviral therapies.
In early July 2005, Pfizer announced that it was to discontinue development of capravirine, after the results of two Phase IIb studies failed to show a statistically-significant difference between standard triple-drug HIV therapies and the same therapy combined with capravirine.
Pfizer remains an established player in the market for antiretroviral drugs with its protease inhibitor Viracept.
NNRTIs ANTIRETROVIRAL DRUGS
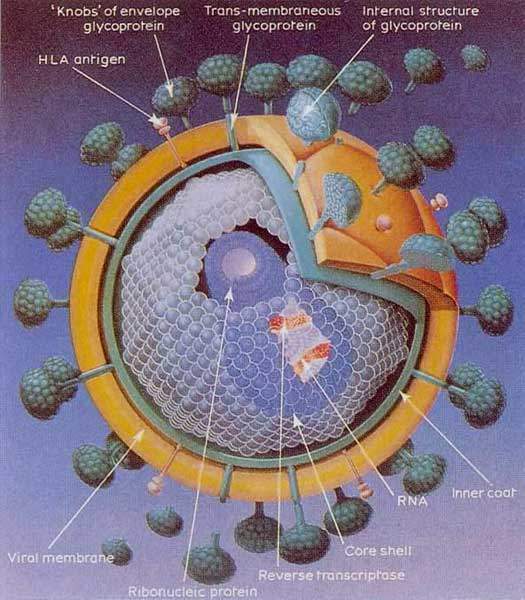
Reverse transcriptase is an enzyme that resides within the cone shaped core of the virus and is essential for viral replication. It is an important target for therapeutic attack. Drugs targeted against this enzyme include nucleoside reverse transcriptase inhibitors (NRTIs) and NNRTIs. The first NNRTI, Viramune (nevirapine), was introduced into clinical practices in 1996. It is indicated for the treatment of HIV-1 infection in treatment-experienced patients with evidence of HIV-1 replication despite ongoing antiretroviral therapy.
NNRTIs work by binding directly to reverse transcriptase, thereby causing structural alterations of the catalytic site. By blocking both RNA- and DNA-dependent polymerase activity they stop viral replication. However, mutations in the enzyme can confer drug resistance to NRTIs and NNRTIs. Preclinical studies with capravirine had shown it had a unique resistance profile. It was active against both wild-type HIV and HIV strains with mutations in reverse transcriptase, including K103N, which leads to the development of broad viral resistance to other commercially available NNRTIs. The emergence of drug resistant strains of HIV is a major factor in treatment failure.
HAART UNDERPINS THE TREATMENT OF HIV INFECTION
The advent of anti-retroviral drugs in the 1990s has had a major impact on the treatment of HIV. While unable to eliminate the virus from the body, these drugs nonetheless reduce symptoms, improve immune status and delay the onset of AIDS. A combination regimen of Highly Active Anti-Retroviral Therapy (HAART) is considered standard treatment for patients with HIV. Ideally, it should be initiated before a patient’s CD4 cell count falls below 350-300cells/ml. Typical regimens include combinations of thymidine nucleoside analogues with a non-thymidine nucleoside analogue. The addition of a protease inhibitor or NNRTI to these regimens may improve outcome.
MID-STAGE CLINICAL TRIALS END CAPRAVIRINE DEVELOPMENT
Capravirine had been subject to a programme of clinical trials, which had focused on two distinct patient populations: treatment-naïve HIV-infected patients and those failing previous treatment regimens.
Evidence from early studies in treatment-naïve patients had suggested that capravirine possessed antiretroviral activity (decreased viral load) when used alone for short periods, as well as achieving target antiretroviral concentrations in plasma as a part of a combination regimen. However, the drug’s failure to meet its efficacy endpoints in subsequent phase IIb clinical trials led to the company’s decision to halt further development.
With the demise of capravirine, Pfizer has just one HIV treatment in late-stage development, its drug maraviroc.
MARKETING COMMENTARY
Since its discovery in the early 1980s, infection with HIV has reached epidemic proportions especially in developing countries. Estimates suggest that 42 million people are living with HIV/AIDS, of whom around 3 million die each year. For HIV patients in industrialised countries with access to treatment, the range of available therapies continues to grow. In the US the market for HIV drugs is expected to more than double by 2011. In addition to new classes of HIV therapies, such as fusion inhibitors, market opportunities exist for new agents in established classes. Because capravirine appears effective against drug-resistant strains of the virus it should become a valuable component of HAART.