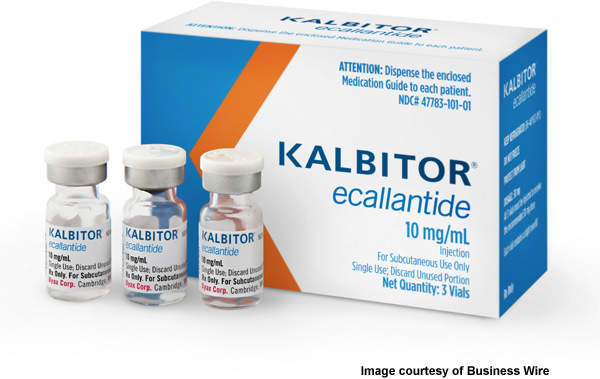
Kalbitor (ecallantide) is a drug developed by US-based Dyax Corp for the treatment of hereditary angioedema (HAE). The drug was discovered using Dyax’s phage display technology. The technology helps sort through antibodies, proteins and enzymes.
The US Food and Drug Administration approved kalbitor in December 2009, making it the second drug to be marketed in the US for treating HAE and the first subcutaneous HAE treatment. Dyax became commercially available in the US in February 2010. In June 2010, Dyax collaborated with Sigma-Tau to develop and commercialise the drug in Europe, Russia, the Middle East and North Africa.
Marketing Authorisation Application (MAA) for approval of Kalbitor in the EU region was submitted to the European Medicines Agency in May 2010. Dyax, however, withdrew the MAA in November 2011 after the Committed for Medicinal Products for Human Use (CHMP) expressed concerns about hypersensitivity reactions in patients treated with the drug. Dyax have stated that they will consider resubmitting the MAA in future.
In December 2010, Dyax extended its collaboration with Sigma-Tau for commercialisation of the drug in Australia and New Zealand. In March 2010, Dyax collaborated with Neopharm Scientific to obtain regulatory approval and commercialisation of Kalbitor in Israel. The company also collaborated with CMIC to develop, regulate and commercialise the drug in Japan, in September 2010.
Hereditary angioedema
HAE, also known as Quincke edema, is a rare heredity disease caused by genetic mutations. The diseases manifests when Cl-esterase-inhibitor (Cl-INH), a naturally occurring molecule, is generated in low or dysfunctional levels.
The basic function of Cl-INH is to regulate plasma kallikrein. Unregulated levels of plasma kallikrein result in excessive generation bradykinin, a vasodilator (a peptide that causes blood vessels to widen) which is responsible for the inflammation and pain caused in HAE attacks. Untreated HAE attacks can last between two to five days and may increase at an unpredictable rate.
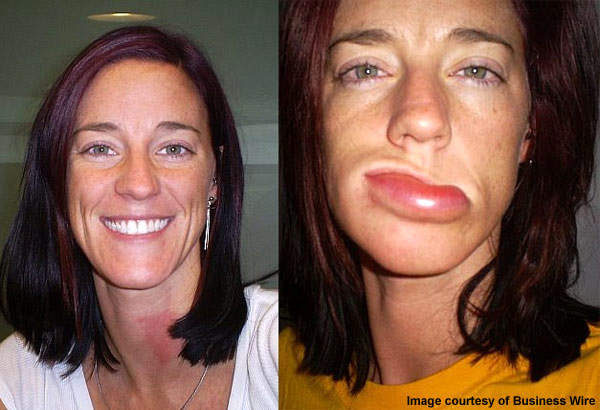
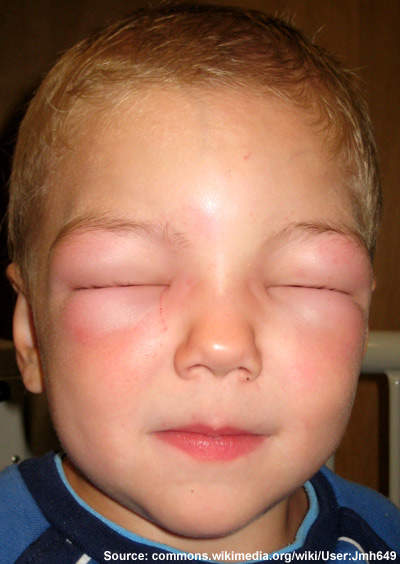
HAE attacks involve painful and severe swelling in various locations of the body. The attacks are irregular and can occur anywhere in the body. The most commonly affected areas include the hands, feet, face, genitals and larynx. HAE attacks involving the larynx are life threatening as they can close off the airway. People affected with HAE continue to have attacks throughout their lives although they may subside with age.
Kalbitor – human plasma kallikrein inhibitor
Kalbitor contains the active ingredient ecallantide, which inhibits the production of plasma kallikrein in HAE patients. During HAE attacks, plasma kallikrein converts high molecular weight kininogen (a protein from the blood coagulation system) into bradykinin. Kalbitor works by binding itself to plasma kallikrein and blocking its binding site. As a result, it prevents the conversion of HMW kininogen to bradykinin.
The mechanism of action of kalbitor provides physicians with a unique approach to treat the unpredictable and severe attacks caused by HAE. In addition, since the drug is administered in the subcutaneous route, its efficacy is higher irrespective of the anatomic location of the attack.
Clinical trials on Kalbitor
The Phase I development programme included four clinical studies on healthy individuals.
Three clinical studies – EDEMA0, EDEMA1 and EDEMA2 – were conducted as part of Phase II development. EDEMA0 was an open-label, escalating, single-dose study which included nine patients experiencing HAE attacks. The second Phase II study, EDEMA1, was conducted in 49 patients who were at least ten years old and experiencing severe to acute attacks of HAE. The studies demonstrated that kalbitor was well-tolerated in the patients.
Approval of kalbitor was based on positive results from two Phase III randomised, double-blind, placebo-controlled studies. The first Phase III study, EDEMA3, involved 72 patients. The studies were conducted across the US, Canada, Europe and Israel. The second Phase III study, EDEMA4, was a randomised, double-blind, placebo-controlled study involving 96 patients across the US and Canada.
In June 2011, around 176 patients were enrolled on a Phase II trial to compare the safety and efficacy of ecallantide with placebo based on conventional therapies. The trial was initiated in patients with angiotension converting enzyme (ACE) inhibitor induced angioedema. It is expected to be completed by November 2011. A Phase II/III trial in paediatric angioedema patients is scheduled to start in October 2011. The trial will evaluate the safety and efficacy of the drug in 18 children aged between two and 17 years. Patients will be intravenously administered either 10-30mg of kalbitor or a placebo. The entire study will be completed by April 2015.
A four-year Phase IV observational study was also started in February 2010 to evaluate the hypersensitivity and immunogenicity of the drug. The study will be conducted on about 200 HAE patients to mitigate any anaphylaxis risks.
Marketing commentary
Before the approval of kalbitor, the only approved drug available to treat acute episodes of HAE was Cinryze, manufactured by Lev Pharmaceuticals for prophylaxis against HAE attacks. Treatment with Cinryze, however, required regular administration of the drug throughout a patient’s lifetime.
In addition, usage of the drug did not eliminate acute attacks. With the absence of a targeted treatment mechanism against HAE attacks, the approval of kalbitor is being considered a significant milestone.
Taiba received the distribution rights of Kalbitor from Dyax in June 2012. Taiba will exclusively distribute the drug in Saudi Arabia, United Arab Emirates, Oman, Bahrain, Qatar, Kuwait, Libya and Iran.
Dyax holds the marketing rights of Kalbitor in the US. The drug was launched in the US market in February 2010.
Novellus Biopharma agreed to partner with Dyax in January 2013 for the development and commercialisation of Kalbitor in Argentina, Brazil, Chile, Colombia, Mexico, and Venezuela.
CVie Therapeutics also entered into a partnership with Dyax in February 2013 for the development and marketing of Kalbitor in China, Hong Kong, and Macau.