Boehringer Ingelheim Pharmaceuticals’ olcegepant (BIBN 4096) is a selective Calcitonin Gene-Related Peptide (CGRP) antagonist, a new class of drugs in development for the treatment of acute migraine attacks.
Olcegepant is undergoing phase II trials in Europe and the US, with preliminary results suggesting that CGRP antagonists may represent a potential new approach to the treatment of migraine.
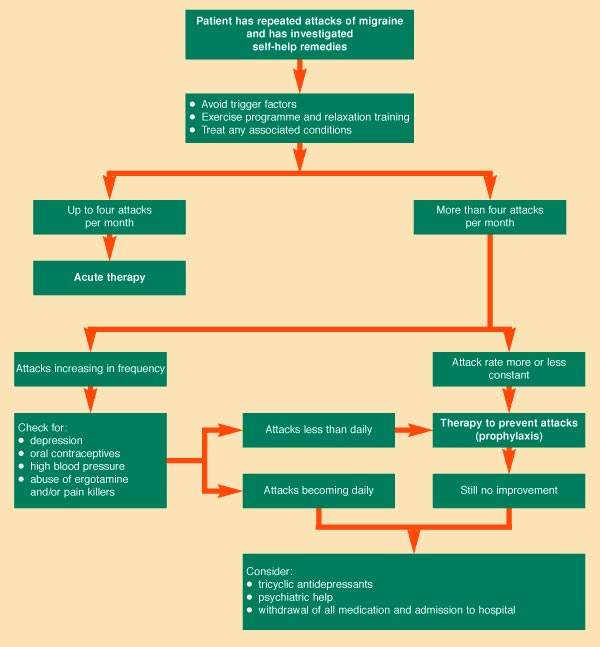
ESTABLISHED MIGRAINE THERAPIES – THE TRIPTANS
The triptans represented a major advance in the treatment of acute migraine when introduced into clinical practice in the early 1990s. They act as selective 5-HT1 agonists and are considered among the most effective agents for
relieving symptoms of an acute migraine attack. Sumatriptan, for example, is often used in the initial headache phase of a migraine attack and to ameliorate other migraine symptoms. It can be administered orally, intranasally, or by subcutaneous
injection and is considered preferred therapy for migraine patients who are refractory to simple analgesics.
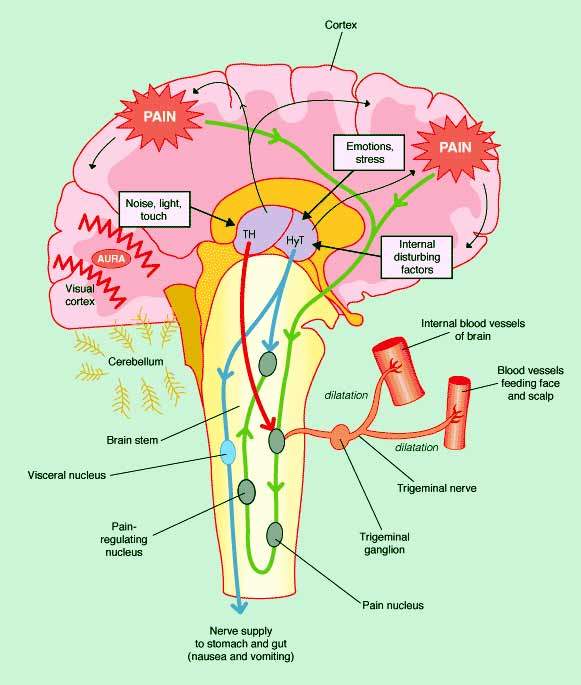
Interestingly, experimental studies have shown that in addition to their primary role as serotonin (1B/1D) receptor agonists, triptans also block the release and action of CGRP, thereby inhibiting trigeminoivascular nociceptive
transmission. Sensitisation and activation of the trigeminal nerves that innervate the meningeal blood vessels is believed to play an important role in the initiation and maintenance of migraine pain. In primary headaches, such as migraine, the release
of CGRP from the trigeminal nerve is closely associated with onset of symptoms.
TOWARDS SELECTIVE CGRP ANTAGONISTS
Although the triptans are highly effective in acute migraine attacks, they are contraindicated in patients with ischaemic heart disease and unstable angina. This has prompted the search for drugs that act selectively as CGRP
antagonists, with potentially fewer cardiovascular side effects than the triptans.
Olcegepant is one of several CGRP antagonists in development as a potential treatment for acute migraine attacks. This new class of drugs is designed to block and/or reverse CGRP-mediated dilation of intracranial vessels induced by
activation of the main sensory nerves in the brain. A highly potent CGRP antagonist, olcegepant has demonstrated the capacity to block release of CGRP in animal models.
PROOF OF CONCEPT DEMONSTRATED IN CLINICAL STUDIES
Building on the results from pre-clinical studies, early clinical trials have since shown that inhibition of CGRP is an effective approach to symptom relief in acute migraine. In a phase II, 126-patient study, intravenous
administration of olcegepant 2.5mg proved significantly more effective than placebo in ameliorating symptoms associated with acute migraine. Among patients receiving olcegepant 2.5mg, the minimum effective dose, 66% of patients responded to treatment
compared with 27% of placebo recipients (P=0.001).
Active treatment was also superior to placebo with respect to secondary efficacy endpoints that included: pain-free at two hours; rate of sustained response over 24 hours; rate of recurrent headache; incidence of nausea,
photophobia, phonophobia, and functional capacity; and time to meaningful relief.
Olcegepant was generally well tolerated. Adverse events occurred in 25% of patients on olcegepant 2.5mg compared with 12% for those on placebo. Paresthesia, a burning or prickling sensation, was the most common adverse event to
occur among olcegepant-treated patients. There were no serious adverse events.
MARKETING COMMENTARY
Affecting up to 10% of the general population, migraine is a common condition. It is classed as a primary headache, a group that also includes cluster and tension-type headaches. First-line treatment of migraine still focuses
primarily on the use of simple analgesics and NSAIDs, although they are not specific anti-migraine therapies and thus carry the risk of rebound headache.
The development of CGRP antagonists as a specific treatment for migraine is still in its infancy but early clinical results are encouraging nonetheless. At present, olcegepant has to be administered intravenously. Developing an equally effective oral
formulation of olcegepant will be important if the drug is to succeed as a new therapy for migraine sufferers.