Vraylar (cariprazine) for the Treatment of Bipolar Disorder and Schizophrenia
Designed for specific mood disorders, Vraylar (cariprazine) is approved for bipolar disorder, schizophrenia and major depressive disorder.

You have successfully submitted your enquiry. Someone from our company will respond ASAP
ACE Pharmaceuticals offers a full range of custom-designed development services with professional standards. Further to that ACE offers manufacturing and packaging of clinical trial medication. We specialize in solid, semi-solid and liquid pharmaceutical products. In a strategic alliance with other companies we can offer you parenteral formulations: ampoules, vials, lyophilised and terminally sterilized processes for single and multi-dose products, small molecules and peptides / proteins and sterility assurance / process validation.
The solutions we offer are tailored to our customers’ exact needs, modular services with the overriding objective of accelerating commercial success. Our expertise covers API characterization, pre-formulation, formulation development, validation and analytical method development. Our GMP-license states that ACE is authorised to manufacture and supply (worldwide) investigational medicinal products for clinical trial purposes.
Our track record includes over 20 products, which have either performed development work, and/or manufactured clinical trial medication, which are now registered products.
Our laboratory is a certified, independently operating analytical laboratory, providing advanced and sophisticated analytical services with experienced professionals and state-of-the-art instrumentation (Certificate 103441 EL, issued by the Ministry of Health). The ACE laboratory has a strong commitment towards quality assurance and provides quality and excellence in analytical support. ACE operates cGMP guidelines and ICH-guidelines.
Our development methods include:
Our service covers the clinical supply process from clinical manufacture to drug destruction in quantities ranging from Phase I and compassionate use requests to large-scale Phase III/IV studies involving multinational centres. It includes packaging, labelling and distribution services for patient administration at investigational sites worldwide. When it comes to driving project completion, ACE Pharmaceuticals provides unsurpassed attention to detail. We deliver always on time, at the right place, in the right quality and the requested quantity for a fair price.
Creation of clinical trial pack design or pack design according to requirement of customer are processed under a number of sections. Our primary packaging includes blinding by over-encapsulation, manufacturing of placebo capsules, blister packaging (PVC, PVDC, PVC/PE/PVDC, Aclar, Aluminium, etc), mixed fill blisters and bottling of tablets and capsules.
Our secondary packaging includes various and complex pack assemblies (open, single blind, double blind, double dummy), cold seal walleting / carding of blisters and coding by inkjet printer (invisible ink).
Finally our labelling services cover the design and generation of labels, multilingual labels, labels printed by thermal transfer, coloured labels or coloured printing, over labelling and design and generation of code break envelopes.
Storage, distribution and returns:
PROJECT MANAGEMENT
Full project management includes randomisation of treatments, protocol development, advice and consultation worldwide; dynamic and GMP/GCP experimented staff.
QUALITY ASSURANCE
Permanent training of operators and the maintenance of our quality system have allowed us to reach an excellent level of quality and to obtain the manufacturer authorisation (license 3103A/d) issued by the Dutch Ministry of Health. This certificate is based on the application of Good Manufacturing Practices and ICH-guidelines dedicated to clinical supplies.

Designed for specific mood disorders, Vraylar (cariprazine) is approved for bipolar disorder, schizophrenia and major depressive disorder.
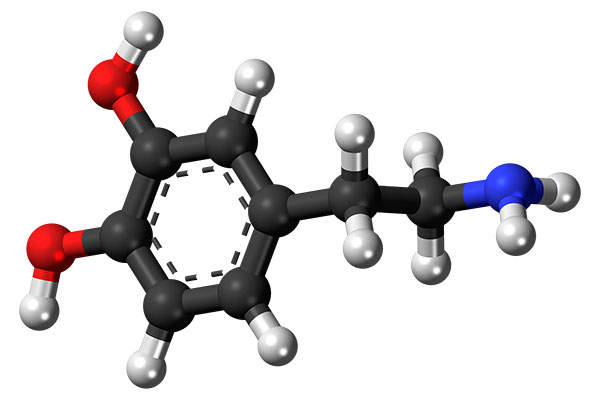
Nuplazid (pimavanserin) is an atypical antipsychotic drug indicated for the treatment of Parkinson’s disease psychosis (PDP).
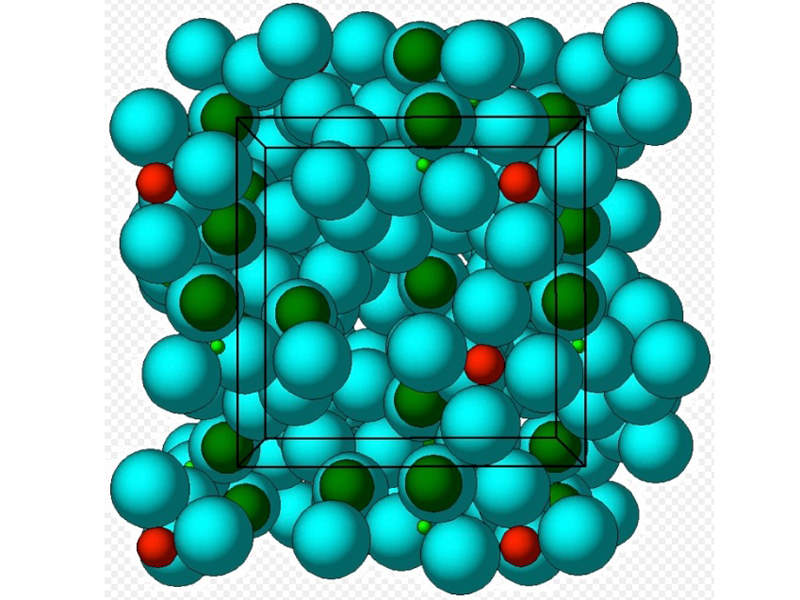
Lokelma is an oral suspension of sodium zirconium cyclosilicate that is indicated for the treatment of patients with hyperkalaemia.
Endari™ is an orally administered powdered form of amino acid L-glutamine indicated for the treatment of sickle-cell disease.
Developed by Eli Lilly and Company, Lartruvo (olaratumab) in combination with doxorubicin is indicated for the treatment of soft tissue sarcoma (STS) in adult patients. The drug is available as a 10mg/ml injection.
MAVYRET™ (glecaprevir/pibrentasvir) was developed by US-based biopharmaceutical company AbbVie as a pan-genotypic treatment for adults suffering from chronic hepatitis C virus (HCV) infection across six major genotypes (GT1 to GT6).
Tecentriq® (atezolizumab) is a monoclonal antibody designed to bind with a protein called PD-L1.
Kanuma™ (sebelipase alfa) is an enzyme replacement therapy (ERT) for the treatment of patients of all ages with lysosomal acid lipase deficiency (LAL-D).

Ninlaro (ixazomib) is a proteasome inhibitor indicated for the treatment of multiple myeloma.
Abilify (aripiprazole), an orally administered dopamine partial agonist and a seratonin antagonist, was approved by US Food and Drug Administration (FDA) in November 2002 for the treatment of schizophrenia.